The Control Experiment
Demonstration
Author: John Taylor Fall 1996 @
HCC-Brandon with Michael and Mary Ann
Background:
In the Fall of 1996, Professor Taylor attempted to perform a
controlled experiment in the Hillsborough Community College Brandon Campus
chemistry lab on a Fall day in 1996. However, everyone was gone on that Friday
afternoon, he had no keys to the stockroom and chemistry technician did not add
all the equipment to the cart. The following items were missing:
1. a Fisher Burner,
2. rubber tubing for the burner,
3. a triple beam balance,
4. Five metal spatulas or scoops,
5. several pieces of glassware.
So the performance is
flawed because additional controls should have been demonstrated.
However, as a student
in your write-up, you should note the places where controls should have been
added if the equipment had been available.
The Chemicals:
The
following Chemicals were used:
A.
Potassium Chlorate KClO3
B.
Sucrose (Table Sugar) C12H22O11
C.
Sodium Chloride (Table Salt) NaCl
D.
Calcium Chloride CaCl2
E.
Barium Chloride BaCl2
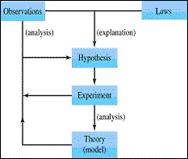
What is the scientific method?
The Experiment (Record Your Notes):
Step 1: Approximate equal amounts of Chemical A and Chemical B were mixed in an evaporating dish and placed on an asbestos pad.
The crystal mixture was lit with a cigarette lighter and a burn occurred:
Your
Observations:
a. What was the appearance and burn
rate of the fire:
b. What did the residue look like:
Step 2: Approximate equal amounts of Chemical A and Chemical B were mixed in an evaporating dish. Then Chemical C was added to the mixture and mixed. The new mixture was placed on another Asbestos Pad.
The new crystal mixture was lit with a cigarette lighter and a burn occurred:
Your
Observations:
c. What was the appearance and burn
rate of the fire:
d. What did the residue look like:
Step 3: Approximate equal amounts of Chemical A and Chemical B were mixed in an evaporating dish. Then Chemical D was added to the mixture and mixed. The third mixture was placed on another Asbestos Pad.
The third crystal mixture was lit with a cigarette lighter and a burn occurred:
Your
Observations:
e. What was the appearance and burn
rate of the fire:
f. What did the residue look like:
Step 4: Approximate equal amounts of Chemical A and Chemical B were mixed in an evaporating dish. Then Chemical E was added to the mixture and mixed. The fourth mixture was placed on another Asbestos Pad.
The fourth crystal mixture was lit with a cigarette lighter and a burn occurred:
Your
Observations:
g. What was the appearance and burn
rate of the fire:
h. What did the residue look like:
Conclusions:
What is a controlled experiment?
What
was the purpose of Chemical A?
What
was the purpose of Chemical B?
What
differences occurred during the four burns:
Burn
#1: A + B + fire
Burn #2: A + B + C + fire
Burn #3: A + B + D + fire
Burn #4: A + B + E + fire
What
were the experimental controls?
What
were variables being tested?
What
was another experimental control incorporated into the variable chemicals when
selecting the compounds to test (C; D; E)?
Suggest
better controls that could have been incorporated into the experiment process
of steps: 1, 2, 3, & 4
Now
conclude a hypothesis or several hypothesi that you
can make after recording your observations:
Hypothesis
I:
Hypothesis
II:
Hypothesis
III
Hypothesis
IV:
Research
flame tests for various cations:
Now
suggest a step #5 for Experiment #1 with a new chemicals:
Step
#5: A + B + F + Flame and using Potassium chloride KCl
as Chemical F
as a fifth chemical to add to the mixture to burn for experiment #1.
From
your research what would you expect to see?
Now
suggest a modification to Experiment #1 by performing an Experiment #2 with a
new set of chemicals that would test your hypothesi:
Burn
#1: A + B + fire
Burn #2: A + B + C + fire Chemical C:_____________________________
Burn #3: A + B + D + fire Chemical D:_____________________________
Burn #4: A + B + E + fire Chemical E:_____________________________
Burn #5: A + B + F + fire Chemical F:_____________________________
If you are ambitious, you form a team of three and perform
Experiment #1 in the FSCJ Chemistry Lab (D-204) with the professor
incorporating additional controls. The processed would be videoed and placed on
the Internet. It would also include Experiment #2 and any additional steps or experiment
to add to the process of using the scientific method.