Project #5: Inorganic
Compound Expert 10 Points
About
Compounds: Nomenclature of Inorganic Compounds
As you become an element
expert in Chapter 3, compounds (Inorganic) are introduced in Section 3.5. Now
it is time for you to become an expert at writing names and formulas of
Inorganic Compounds.
What
is the difference between Organic and Inorganic Compounds?
- All Organic Compounds
contain the element, carbon.
- All Compounds that do not contain the Element Carbon are known as Inorganic Compounds*
*(some compounds such as
carbon dioxide and carbon monoxide are treated as inorganic compounds.)
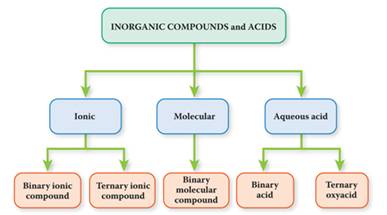
Inorganic
Compounds fall into one of five categories:
How
do I know which category an inorganic compound belongs?
a.
The
key to deciding which category the compound belongs is to look at which element
written first in the compound:
if Metallic then
ionic: Ca3(PO4)2 Calcium
phosphate
if nonmetal then molecular: CO2 Carbon
Dioxide
if hydrogen & (aq) then acid HNO3 Nitric Acid
a. Ionic*(Binary**or
Ternary***): Metallic Cation + Nonmetallic Anion
b. Molecular (Binary**): Nonmetal-Nonmetal
bonded by covalent bonds
c. Acids (aq) (Binary** or Ternary***): Hydrogen + Nonmetallic
anion
(in aqueous solution)
**(Binary Ionic, Binary Molecular,
and Binary Acid only contain two different elements.)
*** (Ternary Compounds contain three or
more different elements)
---------------------------------------------------------------------------------------------------------------------------------------------------------------------
Your first objective is to learn about Elements forming
Compounds in Section 3.4
and viewing the 3.4 Video:
Elements
Can Combine to Form Compounds (two videos)
a. “Compounds Made from Elements” Video #CO304NNa (8:02 Minutes)
You can watch the
video on your cell phone: http://bcove.me/a1rvyyga
b. KM: “Oxygen Bubble Burst” Video
#CO304NNb (3:01 Minutes) (Optional)
You can watch
this video on your cell phone: http://bcove.me/e0jubw52
On Page 73, Figure 3.21
demonstrates the difference between an element and a compound:
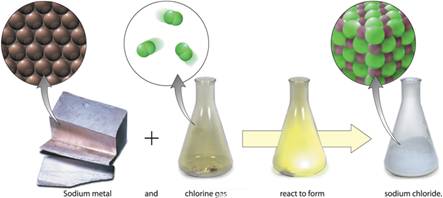
Watch this demonstration video of this
chemical change:
Combination
Demo: SodiumChlorineCombination.mp4
|
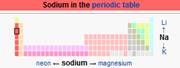
Element:
Sodium (Metal) Sodium /ˈsoʊdiəm/[4] is a chemical
element with symbol Na (from Greek Νάτριο)
and atomic number 11. It is a soft, silver-white,
highly reactive metal.
In the Periodic table it is in column 1 (alkali
metals), and like the other six elements in that column, it has a single
electron in its outer shell that it readily donates, creating a positively
charged atom—a cation. Its only stable isotope is 23Na.
The
free metal does not occur in nature, but must be prepared from compounds as it is highly reactive with water. Sodium is the
sixth most abundant element in the Earth's crust,
and exists in numerous minerals such as feldspars, sodalite and rock salt (NaCl). Many salts of sodium are highly water-soluble:
sodium ions have been leached by the action of water from the Earth's
minerals over eons; sodium and chlorine are the most common dissolved
elements by weight in the oceans. When freshly cut, sodium has a bright, silvery
luster. When exposed to air, the surface rapidly tarnishes, darkening at
first and then forming a white coating of sodium
hydroxide and sodium carbonate. Care is required in handling elemental
sodium because it generates flammable hydrogen and caustic sodium hydroxide on contact with water; powdered
sodium may spontaneously explode in the presence of an oxidizer.[citation needed]
|
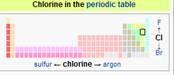
Element: Chlorine (nonmetal) Extremely Toxic! Chlorine is a chemical
element with symbol Cl and atomic
number 17. Chlorine is in the halogen group (17)
and is the second lightest halogen, following fluorine.
The element is a yellow-green diatomic gas under standard conditions.
Chlorine has the highest electron affinity and the third highest electronegativity of all the reactive elements.
For this reason, chlorine is a strong oxidizing
agent. Free chlorine is rare on
Earth, and is usually a result of direct or indirect oxidation
by oxygen. The most common compound of chlorine, sodium
chloride (common salt), has been known since ancient times. Sir
Humphry Davy in 1810 named it from Ancient Greek:
λωρός (khlôros) "pale
green". Nearly all chlorine in the Earth's
crust is in the form of ionic chloride compounds, which includes table salt. It is the second most abundant halogen and
21st most abundant chemical element in Earth's crust. Elemental chlorine at high concentrations is extremely dangerous and poisonous for all livingorganisms.
|
Compound: Life
Giving!
|
So, two extremely dangerous elements combine to form a compound
essential for life!
-------------------------------------------------------------------------------------------------------------------------------------------------
Another
Example:
|
Compound:
NaCl(s) Life Giving
|
Element:
Life Giving |
= |
New
Compound: Sodium
Hypochlorite NaClO (s) Extremely Toxic |
Just varying the composition
of a compound by one element or even one atom changes
the compounds chemical properties
-------------------------------------------------------------------------------------------------------------------------------------------------------
Another
Example:
Look Mom No Cavities!
|
|
|
|
In
1954 Crest Toothpaste was born. It was
the first tooth paste with
fluoride to prevent cavities.
Hoverer, Crest advertised that their tooth paste contained Stannous
fluoride. But they did not use the stock
name, Tin
II Fluoride*, because the word Tin would be misunderstood as the element Tin, a shinny
metal
Look Mom No Cavities!
[Tin(II) fluoride, commonly referred to commercially (with
tin(II) in Latin)
as stannous fluoride,is a chemical
compound with the formula SnF2. It is a
colorless solid used as an ingredient in toothpastes
that are typically more expensive than those that use sodium
fluoride. Stannous
fluoride converts the
calcium mineral apatite into fluorapatite, which
makes tooth
enamel more resistant to bacteria-generated acid attacks. In
toothpastes containing calcium minerals, sodium fluoride becomes ineffective
over time, while stannous fluoride remains effective in strengthening tooth
enamel.[3]
Stannous fluoride has been shown to be more effective than sodium
fluoride in reducing the incidence of dental caries[4]
and controlling gingivitis].
[Stannous
fluoride is used under the trade name "Fluoristan" in the original formulation of the toothpaste Crest, though it was later replaced with sodium monofluorophosphate,
or "Fluoristat". It is the active
ingredient in Crest Pro Health brand toothpaste. Crest Pro Health issues a
warning on the tube that stannous fluoride may cause staining, which can be
avoided by proper brushing, and that its particular formulation is resistant to
staining. Any stannous fluoride staining that occurs due to improper brushing
is not permanent. Stannous fluoride is also used in Oral-B Pro-Expert.[6] Stannous fluoride is also readily available in
over-the-counter rinses.]
---------------------------------------------------------------------------------------------------------------------------------------------
Your next objective is to learn to write the Names and Formulas
of Inorganic Compounds in Section 3.5 by viewing the 3.5 Video:
There
Is A System for Naming Compounds
a.””Naming
Compounds” (only Binary Ionic and
Binary Molecular discussed)
Video #CO305a
(3:30 Minutes)
Watch
the video on your cell phone: http://bcove.me/25ylz1i9
--------------------------------------------------------------------------------------------------------------------
Your next objective is to learn to write the Names and Formulas
of Inorganic Compounds in Section 3.5 by applying the riles from the Guidelines lised in the power point for chapter 3 or by viewing study
guides highlighted in yellow below.
Complete all 12
Tasks for Names and Formulas of Inorganic Compounds for 9 points (120 total
compounds-10 compounds/task) and you must score 8 or more out of 10 to
successfully complete the task.
Record
and Submit your scores on the
Project #5 Online Nomenclature Verification
Form
Guideline #1 Task #1: Binary
Ionic Compound Names Online Exercise
Guideline #1 Task #2 Binary
Ionic Compound Formulas Online Exercise
Binary
Ionic Online Study Guide
Guideline #2 Task #3 Binary
Molecular Compound Names Online Exercise
Guideline #2 Task #4 Binary
Molecular Compound Formulas Online Exercise
Binary
Molecular Online Study Guide
Guideline #3 Task #5
Polyatomic
Ion Names Online Exercise
Guideline #3 Task #6 Polyatomic
Ion Formulas Online Exercise
Polyatomic
Ion Online Study Guide Partial Polyatomic Ion
List Complete
List
For the last six tasks you should have
the polyatomic ion list at your fingertips:
Guideline #3 Task #7
Ternary
Ionic Compound Names Online Exercise
Guideline #3 Task #8 Ternary
Ionic Formulas Online Exercise
Ternary
Ionic Online Study Guide
Guideline #5 Task #9
Binary/Ternary
Acid Compound Names Online Exercise
Guideline
#5 Task #10 Binary-Ternary
Acid Compound Formulas Online Exercise
Binary-Ternary
Acid Online Study Guide
Guidelines #1, #2, #3, & #5 Task #11 Inorganic
Compound Names Online Homework
Guidelines #1, #2, #3, & #5 Task #12 Inorganic
Compound Formulas Online Homework
Inorganic
Compound Online Study Guide
----------------------------------------------------------------------------------------------------------------------------------------------
MSDS Research (Material Safety Data Sheet) 1 point
When we purchase chemical there is data sheet which is
packaged with the chemical, like the poop sheet
which is included in a prescription
drug. This data must be kept by law in our stockroom for every chemical on the
shelf
These data sheets are called the MSDS Guide (Material
Safety Data Sheet). You assignment for 1 point is research one chemical :
|
1. calcium chloride 2. sodium sulfate 3. barium chloride 4. hydrochloric acid 5. sulfuric acid 6. nitric acid 7. naphthalene 8. sucrose 9. potassium chloride 10. silver nitrate 11. sodium nitrate 12. ethanol (ethyl alcohol) 13. nickel(II) sulfate 14. copper(II) sulfate 15. magnesium sulfate |
16. iodine 17. magnesium 18. magnesium oxide 19. 2-propanol
20. sodium hydroxide 21. ammonia 22. aluminum chloride 23. magnesium chloride 24. boric acid 25. phosphoric acid 26. ascorbic
acid |
27. silicon dioxide (sand) 28. camphor 29. sodium bicarbonate 30. sodium acetate 31. methyl alcohol (methanol) 32. formaldehyde (Methanal) 33. Benzene 34. acetic acid (Ethanoic acid) 35. sodium carbonate 36. Stannous fluoride 37. Sodium 38. Acetone 39. Sodium borate (Borax) 40. Boric Acid |
Some chemical information sites to
research your assigned chemical:
Iowa State University: http://avogadro.chem.iastate.edu/MSDS/MSDS_P.html
MSDSexchange: http://www.msdsxchange.com/english/index.cfm
-------------------------------------------------------------------------------------------------------------------
Sample
MSDS for a Chemical: Acetone

---------------------------------------------------------------------------------------------------------------------------------------------------
MSDS
Laboratory Assignment
(Please submit your form with this assignment Place numbers in labels
below.)
NFPA Codes
HMIS Codes

Student Name: ____________________Class Section:______
Compound: ________________________________
Chemical Formula: _________________________
Appearance & Odor:
________________________
Boiling Point: _______ Melting Point: ________
Solubility in water:
__________________________
Conditions to Avoid:_________________________
___________________________________________
Materials to Avoid: __________________________
Using
Chemical Date Bases/Web Search to fill in the above form and submit as an
attachment to an email when Project #5 is complete.
Safety Codes
Hazard
Codes
ChemAlert rates hazards numerically inside the NFPA (National Fire Protection Association) diamond. This symbol was chosen for its universality.
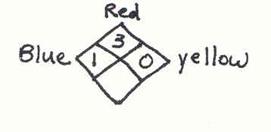
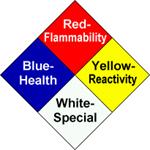
The diamond has a red segment (flammability), a blue
segment (health, i.e. toxicity), a yellow segment (reactivity),
and a white/blank segment (special warnings such as radioactivity or no
water). Printed over each is a bold
black number expressing the degree of the hazard.
The
numerical ratings are:
4 = extreme hazard
3 = severe hazard
2 = moderate hazard
1
= slight hazard
0 = according to present data, none
Storage Codes
The storage code is
assigned according to the chemicals worst hazard:
RED Flammable
YELLOW Reactive
and Oxidizing Agents. May react violently with air,
water, or other
substances.
BLUE Health Hazard. Toxic if inhaled, ingested, or absorbed
through
the skin.
WHITE Corrosive. May harm skin, eyes, or
mucous membrane.
GREEN or Presents no more than moderate hazard in any category.
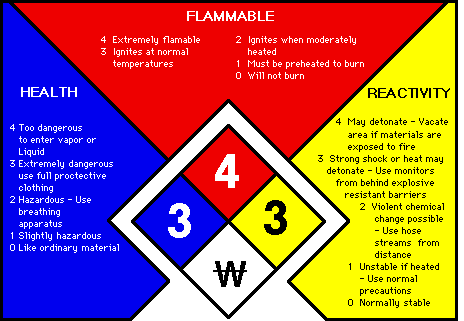
Link to the following Government web site for more details:
http://www.sefsc.noaa.gov/HTMLdocs/nfpahazard.htm
This site has the following symbol


HEALTH - The degree of health hazard of a chemical or material is based on the form or condition of the material, as well as its inherent properties. The degree of health hazard of a material should indicate the degree of personal protective equipment required for working safety with the material.
1 is for slightly
hazardous (toxic) material which requires only minimal protection (for example,
safety glasses and gloves) in addition to normal work clothing to work with
safely.
2 is for
moderately toxic or a hazardous or moderately toxic material which requires
additional PPE or equipment (e.g. chemical goggles, lab/work smock, local
ventilation) in addition to that required for less toxic material. Consult the
MSDS for specific health hazard and proper PPE to use with this material.
3 or 4 is for highly to extremely toxic (deadly) materials (and any
carcinogen, mutagen, or teratogen). These materials will require specialized
equipment (e.g. respirator or exhaust hood, full face shield, rubber apron,
specialized glove, handling tongs, etc) beyond that required for moderately
toxic material. You must consult the
MSDS and/or other safety information to determine the hazard (acute or chronic)
and the proper PPE and engineering controls to safely use this material.
FLAMMABILITY or FIRE HAZARD - The flammability or fire hazards deal with the degree of susceptibility of the material to ignite and burn. The form or condition of the materials, as well as their properties, affects the extent of the hazard. Many hazardous materials such as acetone and gasoline, have a flash point (ignition temperature) far below freezing and will readily ignite with a spark if the vapor concentration is sufficient.
1 is for
materials with a flash point above 200ºF.
2 is for materials with a flash point below 200ºF but above 100ºF.
3 is for materials with a flash point below 100ºF but above 73ºF.
4 is for materials with a flash point below 73ºF.

REACTIVITY - The reactivity hazards deal with the potential of a material or chemical to release energy. Some materials are capable of rapid energy release without any catalyst, while others can undergo violent eruptive or explosive reactions if they come in contact with water or other materials. Generally this rating is used to indicate the potential to react if the material is heated, jarred, or shocked.
1 indicates a
material that may be reactive if heated and one that reacts with water.
2 indicates a material that may react violently without
detonation.
3 indicates a material that may detonate or explode if
subjected to a strong initiating force or heating under confinement.
4 indicates a material that readily detonates or
explodes.
SPECIFIC HAZARD - An open space at the bottom of the NFPA diagram can be used to indicate additional information about the chemical or material. This information may include the chemical or material's radioactivity, proper fire extinguishing agent, skin hazard, its use in pressurized containers, protective equipment required, or unusual reactivity with water.
OX or OXY
indicates a material that is an oxidizer.
W or W indicates a material that is water reactive.
ALK indicates a
material that is alkali.
COR indicates a
material that is corrosive.
RAD indicates a material that is radioactive.
Special Labeling Requirements
All containers
that hold carcinogens, reproductive hazards or acutely toxic chemicals must be
properly labeled concerning the health hazard posed by the chemical. Most containers will have the chemicals
hazard clearly displayed on the label.
However older chemicals and containers of solutions that are mixed in
the lab must be properly labeled by the laboratory worker. The laboratory worker may write the hazard
class (e.g. carcinogen, etc.) on the container or use labels available from
their Supervisor or Chemical Hygiene Officer.
Symbols You Should be Familiar:


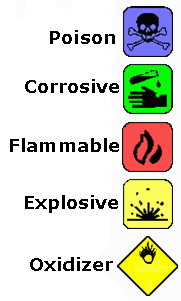








-----------------------------------------------------------------------------------------------------------------------
Hazardous Materials Identification System (HMIS)
Lab Symbols You Should
Know:



Don’t Confuse HMIS with NFPA Labeling. Read:
http://www.ilpi.com/Msds/ref/hmis.html








The MSDS web site may be accessed at the following:


http://www.ilpi.com/msds/ref/nfpa.html
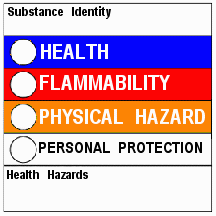
Below is a paragraph about the two labeling systems:
“At first glance, the HMIS® and NFPA labeling systems appear quite similar. Both have four sections colored blue, red, yellow and white. HMIS® uses colored bars, while NFPA uses colored diamonds. HMIS® attempts to convey full health warning information to all employees while NFPA is meant primarily for fire fighters and other emergency responders.”
.
MSDS Relevance
Specific sections of an HMIS® label include the following:
|
Health |
|
|
o
The Health
section conveys the health hazards of the material. In the latest version of
HMIS®, the blue Health bar has two spaces, one for an asterisk and one for a
numeric hazard rating. If present, the
asterisk signifies a chronic
health hazard, meaning that long-term exposure to the material could
cause a health problem such as emphysema or kidney damage. NFPA
lacks this important information because the NFPA system is meant only for
emergency or acute
(short-term) exposures. According to NPCA, the numeric hazard assessment procedure is different than that used by NFPA. Here are the numeric rankings for the HMIS system: |
|
|
4 |
Life-threatening, major or
permanent damage may result from single or repeated overexposures. |
|
3 |
Major injury likely unless
prompt action is taken and medical treatment is given. |
|
2 |
Temporary or minor injury
may occur. |
|
1 |
Irritation or minor
reversible injury possible. |
|
0 |
No significant risk to
health. |
|
|
|
|
|
|
|
Flammability |
|
|
For HMIS I and II, the criteria used to assign numeric values (0 = low hazard to 4 = high hazard) are identical to those used by NFPA. In other words, in this category, the systems are identical. For HMIS III, the flammability criteria are defined according to OSHA standards: |
|
|
4 |
Flammable gases, or very volatile flammable
liquids with flash
points below 73 °F,
and boiling points
below 100 F. Materials may ignite spontaneously with
air. (Class IA) . |
|
3 |
Materials capable of ignition under
almost all normal temperature conditions. Includes flammable liquids with
flash points below 73 °F
and boiling points
above 100 °F, as well as liquids with flash points between
73 °F and 100 °F. (Classes IB & IC). |
|
2 |
Materials which must be
moderately heated or exposed to high ambient temperatures before ignition
will occur. Includes liquids having a flash point at or
above 100 °F but
below 200 °F. (Classes II & IIIA). |
|
1 |
Materials that must be
preheated before ignition will occur. Includes liquids, solids and semi
solids having a flash
point above 200 °F.
(Class IIIB). |
|
0 |
Materials that will not
burn. |
|
Physical Hazard (HMIS® III) |
|
|
o
Reactivity hazard
are assessed using the OSHA
criterion of physical
hazard. Seven such hazard classes are recognized: §
Compressed
gases §
Pyrophoric
materials. §
Unstable Reactives |
|
|
4 |
Materials that are readily capable of explosive water reaction,
detonation or explosive decomposition, polymerization, or
self-reaction at normal
temperature and pressure. |
|
3 |
Materials that may form explosive mixtures with water and are
capable of detonation or explosive reaction in the presence of a strong
initiating source. Materials may polymerize, decompose,
self-react, or undergo other chemical change at normal temperature and pressure
with moderate risk of explosion. |
|
2 |
Materials that are unstable
and may undergo violent chemical changes at normal temperature and pressure
with low risk for explosion.
Materials may react
violently with water or form peroxides upon exposure
to air. |
|
1 |
Materials that are normally
stable but can become unstable (self-react) at high temperatures and
pressures. Materials may react non-violently with water or undergo hazardous polymerization
in the absence of inhibitors. |
|
0 |
Materials that are normally
stable, even under fire conditions, and will not react with water, polymerize, decompose ,
condense, or self-react. Non-explosives. |
|
Personal Protection |
|||||||||||||||||||||||||||
|
o
This is by far
the largest area of difference between the NFPA and HMIS® systems. In the
NFPA system, the white area is used to convey special hazards
whereas HMIS® uses the white section to indicate what personal protective equipment
(PPE) should be used when working with the material. Note: The NPCA specifically recommends that "preparers of MSDSs should not place HMIS® PPE
designation codes on the MSDSs or labels that leave the facility, as they
do not know the conditions under which their customers use those products."
However, these still turn up on some MSDS's. HMIS® uses a
letter coding system for this section. We at ILPI find this unacceptable
because we would rather see the PPE listed explicitly instead of having
employees try to remember a bunch of codes or consult a chart, something that
could lead to confusion and/or a fatal accident. Likewise, the "custom
codes" aspect is particularly dangerous for visitors and contractors who
may not remember/recognize that these could vary from job site to job site.
We
present the lettering scheme here, along with a series of graphics meant to
reinforce the meaning of each letter:
|
As a lay person do not let an employer make you handle a chemical on the job without the proper protection. This exercise to search a data base for information about a chemical and its hazards should be taken seriously. Before you search the chemical data base to look up the MSDS data, Google the chemical and read the general information about the chemical such as presented above in some the examples above.