CHM
1025C Name: _________________
Module
Five I Homework Packet-Hein
Module 5i: Chemical
Equations & Stoichiometry (Chapter 8)
E._____(02) Writing Reactions/ Symbols-Section 8.1 Answers
E1.____(02) Classifying Chemical Reactions- Section 8.3 Answers
F._____(05) Balancing Chemical Equations -Sections 8.2 Answers ef
G._____(04) Predicting Single Replacement Products . Sections 8.3 Answers
H._____(04) Predict Double Replacement Sections 8.3 Answers h
H1____ (04) Neutralization/Gas Forming Reactions 15.6 ,Lecture Answers
______(21)
Module 5i Total (Ninth Exam)
Foundations of College Chemistry, 14th Edition
Table of Contents
Chapter 8 Chemical
Equations 143
8.1
The Chemical Equation 144
8.2
Writing and Balancing Chemical Equations 145
8.3
Types of Chemical Equations 150
8.4
Heat in Chemical Reactions 156
8.5
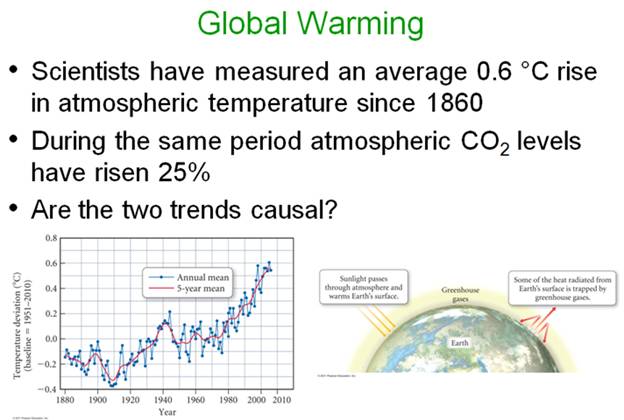
Global Warming: The Greenhouse Effect 159
Review
161
Review
Questions, Paired Exercises 163
Additional
Exercises 165
Challenge
Exercise, Answers to Practice Exercises 166
In Module Three
we studied the composition of an atom.
In Module Four
we made compounds.
In Module Five
we finally should be able to react
these compounds in
chemical reactions.
(or summarize a chemical
change we studied in Module 1).
From Your Hein Textbook and Power Point for
Chapter 8, what is a chemical equation?




Evidence for
Chemical Reactions
There are four observations that indicate a
chemical reaction is taking place:
1.
A gas is produced.
Gas may be observed in
many ways in a reaction from light fizzing to heavy bubbling.
2.
An
insoluble solid is produced
in a solution.
a. A substance
dissolves in water to give an aqueous solution.
b. If we add
two aqueous solutions together, we may observe the production of a solid
substance.
c. The
insoluble solid formed is called a precipitate
3. A permanent color change is observed.
a. Many
chemical reactions involve a permanent color change.
b. A change in
color indicates that a new substance has
been formed
4. An energy change is observed
a. A reaction
that releases heat is an exothermic reaction.
b. A reaction
that absorbs heat is an endothermic reaction.
c. Examples of
a heat energy change in a chemical reaction are heat and light being given off.
From the Corwin Textbook, the Chemical symbols are explained:

M-5i Homework Packet
Module
Five-Part E Basic Stoichiometry
Definitions 2 points
Fill in the
following with the symbols used in chemical equations which has the stated
translation or meaning(s) (Corwin Section 7.2 Table 7.1 page 191 Corwin 7th ) (Hein Section 8,1 Table 8.1 page 144):
_________1.
Produces, yields, gives
_________2.
Reacts with, added to, plus
_________ and _________ 3. Solid substance or precipitate forms
_________and _________4. Gaseous substance formed
_________5.
Liquid Substance
________5a. Water or aqueous solution
_________6. Reversible Reaction
_________7.
No Reaction
8. Show the
symbol for heat:__________
9. How
would you show a catalyst in a chemical reaction where A plus B forms products
D and E, but is catalyzed by substance C
A + B à D + E
10. Define
Catalyst (See Section 7.2 page 191)
M-5i Homework Packet
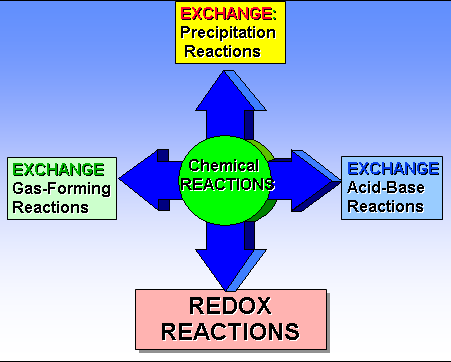
In Corwin’s 6th Section 8.4 five types of reactions: Combination, Decomposition, Single Replacement, Double Replacement, and Neutralization reactions are introduced. The table 8.3 is a summary. Hein Section 8.3 explains the five types.

In Hein’s 14th edition Section 8.3 is devoted to types of chemical reactions, pages 150-156 which summaried in Chapter 8 Power Point:

From my lecture, I have told you a chemical change via a chemical reaction is either a NON-REDOX or a REDOX change. Combination, Decomposition, Single Replacement and Oxidation-Reduction (more complicated) are REDOX changes. Only Double Replacement, Double Displacement or sometimes called Metatarsus reactions are NON-REDOX and I like to call them Ion-Exchange Reactions,.
Our Hein
textbook describes several types of Double Displacement Reactions including gas
formation. Our Corwin textbook should have a section 7.12 which is the third
type of double replacement (Metatarsus or Ion Exchange):
Gas Forming Reactions.
When
predicting the products of a double replacement reaction, sometimes one of the
products instantly decomposes.
If H2CO3
is a predicted product in ion exchange, it is written as
CO2 and H2O.
For example:
Na2CO3
(aq) + HCl (aq)
à [H2CO3](aq)
+ NaCl (aq)
Should
be written:
Na2CO3
(aq) + HCl (aq)
à CO2(g) + H2O
(l) + NaCl (aq)
Two
other products which are shown differently:
[NH4OH] à NH3 + H2O
[H2SO3 ] à SO2 + H2O
In Corwin’s
chapter 19 and Hein’s Chapter 20, there is a another
type of chemical reaction:
Combustion – a substance burns in the
presence of oxygen. Combustion of a compound that contains C and H (or C, H,
and O) produces carbon dioxide gas and water.
CH2O(l) + O2(g)
→ CO2(g) + H2O(l)
The general type of combustion problem looks
like this:
CxHy
[(l) or (g)] + O2 (g) à CO2 (g) + H2O
(g)
Or
CxHyOz[(s) or (l) or
(g)] +
O2 (g) à CO2 (g) + H2O
(g)
The
chemical reaction for the combustion of gasoline:
C8H18
(l) + O2 (g) à CO2
(g) +
H2O (g)
Octane oxygen gas carbon dioxide water
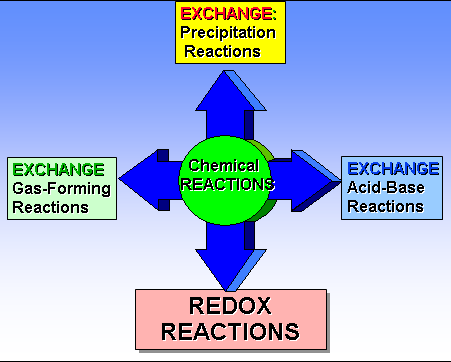
Module 5 E1: Classification of Chemical Reactions 2 Points
(Sections 7.4)
Classify Each of the
following (unbalanced) chemical reactions as:
- Combination
(or synthesis)
- Decomposition
(or Anaylsis)
- Single
Replacement
- Double
Replacement (Precipitation)
- Double
Replacement (Neutralization)
- Double
Replacement (Gas Forming)
- Combustion
of a hydrocarbon
______1. Fe
+ FeCl3 à FeCl2
____2. HCl + Mg(OH)2 à MgCl2 +
HOH
_____3. Mg +
HNO3 à Mg(NO3)2 +
H2
_____4. H2 + N2 à NH3
____5.
NaHCO3 + HCl
à
NaCl + CO2 + H2O
____6.
Ca(NO3)2 + K3PO4 à Ca3(PO4)2 + KNO3
____7.
KClO3 à KCl + O2
____8.
Na + H2O à NaOH
+ H2
(See Corwin’s
Questions #21-24 p215
for more practice) (Hein has no additional practice at the end of the chapter)
Writing
Chemical Reactions (Section 7.2)
9.
Write a chemical equation for solid cadmium hydrogen carbonate decomposing to
yield solid cadmium carbonate, water, and carbon dioxide gas:
10.
Write a chemical equation for the reaction of aqueous solutions of potassium
chromate and calcium sulfate to give the precipitate calcium chromate and
aqueous potassium sulfate.
See
additional Corwin’s questions #7-17 p215;
Hein Questions #9-#12 page 164.



Writing
Chemical Reactions
11.
Write a chemical equation or solid sodium hydrogen carbonate decomposing to
yield solid cadmium carbonate, water, and carbon dioxide gas:
12.
Write a chemical equation for the reaction of aqueous solutions of potassium
chromate and lead(II) nitrate to give the precipitate
lead(II) chromate and aqueous potassium nitrate.
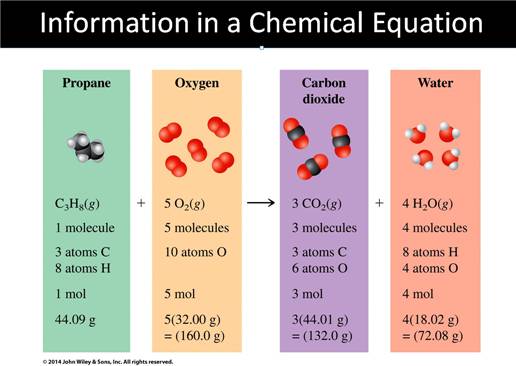
Rules and Suggestions
for Balancing Equations
1)
The same # and
type of atom must be present on each side of the equation.
2)
Balancing is
accomplished by adding coefficients. NEVER change the subscripts.
3)
Coefficients must
be in the smallest whole # ratio.
4)
Balancing is done
by trial and error.
5)
Usually Balance
H’s and O’s last or an element that appears in more than one place of either
side of the reaction .
6) Balance polyatomic ions as one unit in Ion Exchange reactions.


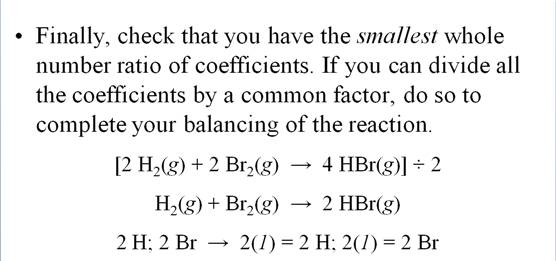
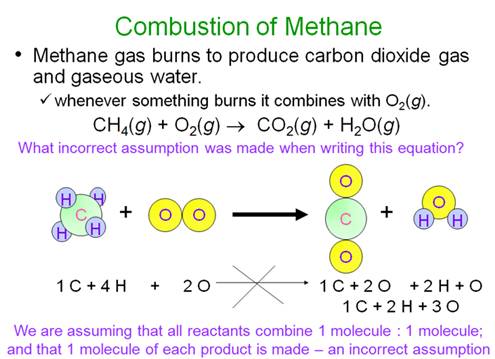
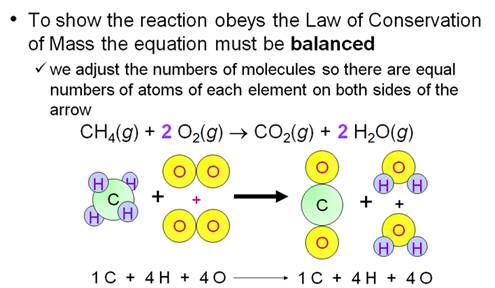
M-5i Homework Packet
Example of balancing an Equation:


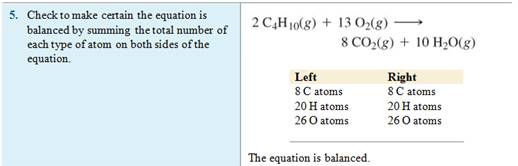
Module
Five-Part F Balancing Chemical Equations
5 points
Balance the
following chemical equations (write the chemical formulas in #10 then balance):
1. Fe
+ FeCl3 à FeCl2
2. Al +
O2 à Al2O3
3. Na2CO3 +
C + N2 à NaCN
+ CO
4. FeS + O2 à Fe2O3 +
SO2
5. IBr +
NH3 à NI3 + NH4Br
6. Cl2 +
HOH à
HCl + HClO
7. AgNO3 à AgNO2 +
O2
8. HClO4 +
P4O10 à H3PO4 +
Cl2O7
9. HCl + Mg(OH)2 à MgCl2 +
HOH
10.
Sodium hydroxide + Hydrochloric
acid à sodium chloride +
water
Hein
Additional Practice: Page 163 Questions #3-#8
You
may check your work using the online chemical equation balancer at:
http://people.emich.edu/bramsay1/ccc-release/chem.html
M-5i Homework Packet

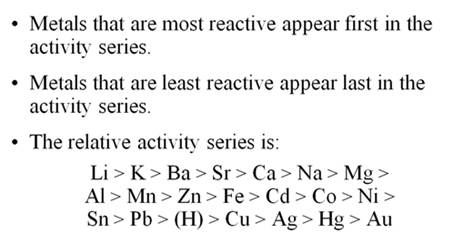

M-5i Homework Packet


M-5i Homework Packet


See
Sections 7.7 and 7.8. Extra practice
questions on page 216 #47-52; and #53-55 page 217.
M-5i Homework Packet
If you were to (H2O) in the activity
series like an acid is shown as (H), where would you put it? Show below:
Given
the following Activity Series:
Li > K
> Ba > Sr > Ca > Na >
Mg > Al >
Mn > Zn
> Fe > Cd > Co > Ni >
Sn >
Pb > (H)
> Cu > Ag
> Hg > Au
The rule to follow is a single replacement reaction takes place only if the metal or (H) is more active than the metal or (H) it is replacing. Li will react with everything, while Hg will replace only gold. And poor gold does not react with any of the cations of metals. Therefore gold is found pure in nature, while the very active metals such as potassium and sodium are never found pure in nature, but are found as minerals (ionic compounds).
Will Mg metal react with Nitric Acid?
Yes
(Mg has a great
reactivity then [H] in the series)
Mg (s) + 2 HNO3 (aq) → Mg(NO3)2 (aq) + H2 (g)
Will Copper react with Nitric Acid? no
(Cu is below [H] in
the activity series)
Cu (s) + HNO3 (aq) → no reaction
Given the following Active Metals:
Li > K > Ba > Sr > Ca > Na> (H2O)
The six very active metals are so reactive they will replace one of the two hydrogens in water and form alkaline hydroxides as products. Hydrogen gas will bubble out of the solution. See some of the above movies for demonstrations.
Will Sodium react with water? yes
(Na is one of the six
active metals above)
2 Na(s) + 2 HOH (l) → 2 NaOH (aq) + H2 (g)
M-5i Homework Packet
Part
G Single Replacement Reactions 4 points
Given
the following Activity Series:
Li
> K > Ba > Sr > Ca > Na > Mg > Al > Mn > Zn > Fe
> Cd > Co > Ni >
Sn
> Pb > (H) > Cu > Ag > Hg > Au
Given
the following Active Metals:
Li
> K > Ba > Sr > Ca > Na
Complete
the products of the following reactions, then balance the equation (If no
reaction write NR):
1. Cu (s) + Al(NO3)3 (aq) à
2. Al (s) +
Cu(NO3)2 (aq) à
3. Au (s)
+ H2SO4
(aq) à
4. Ca (s) +
H2O (l) à
5.
Mn (s) + H2O (l) à
For additional Practice:
Hein page 164 Questions #13-#16
M-5i Homework Packet
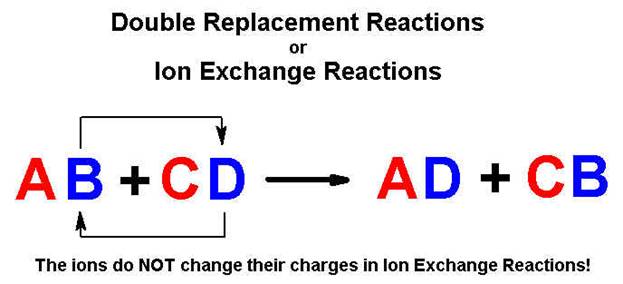

Except in neutralization and gas forming reactions.

Part
H Double Replacement Reactions 4 points
Given the following Solubility Rules for Ionic
Compounds:
Compounds containing the following ions are
generally soluble in water:
1. Alkali metal ions and ammonium ions, Li+ , Na+ , K+ , NH4+
2. Acetate ion, C2H3O2-
3. Nitrate ion, NO3-
4. Halide ions (X), Cl- , Br-
, I- (AgX, Hg2X2 ,
and PbX2 are insoluble
exceptions)
5. Sulfate ion, SO4 2-
(SrSO4, BaSO4
, and PbSO4 are
insoluble exceptions)
Compounds containing the following ions are
generally insoluble in water:
6. Carbonate
ion,CO32- (see rule 1
exceptions which are soluble)
7. Chromate
ion CrO42- (see rule 1 exceptions which are soluble)
8. Phosphate ion PO43-
(see rule 1 exceptions which are soluble)
9. Sulfide
ion, S2- (CaS, SrS, BaS, and rule 1
exceptions are soluble in water)
10. Hydroxide ion, OH- [ Ca(OH)2
, Sr(OH)2 , Ba(OH)2 , and rule 1 exceptions are soluble)
Complete and balance the following reactions using
the above solubility table (write no reaction or NR if both products are
soluble or a covalent compounds is not formed)
1. AlCl3
(aq) + K2CO3 (aq) à
2. NiSO4
(aq) + Li3PO4 (aq) à
3. NaCl
(aq) + AgNO3 (aq) à
4. H2SO4
(aq) + NaOH (aq) à
5. H3PO4
(aq) + Ba(OH)2 (aq) à
Video:
http://www.brightstorm.com/science/chemistry/chemical-reactions/double-replacement-reactions/
http://www.youtube.com/watch?v=7hVKb4ROjZw
http://www.youtube.com/watch?v=_oixjNeKtxs
http://www.youtube.com/watch?v=IMfNi_C2DTg
http://www.youtube.com/watch?v=2tIutF6-wn4
See
Corwin’s Sections
7.9 and 7.10. Hein’s section 8.3 pages 153-156.
Additional Practice Problems:
Corwin Page 218 #71-74; Hein Page 164 #11 and #12;
page 165 #39-40
M-5i Homework Packet

Part
H1 Double Replacement Reaction:
Neutralization/Gas Forming Reactions 2 points
Complete and balance the following precipitation
reactions using the above solubility table
(write no reaction if both
products are soluble or a covalent compounds is not formed)
1. Mg(OH)2 (s)
+ H2SO4
(aq) à
2. H3PO4
(aq) + KOH (aq)
à
3. NH4NO3
(aq) + Ba(OH)2 (aq) à
4. HBr (aq) + Pb(CO3)2 (aq) à
5. LiOH
(aq) + H3PO4 (aq) à
6. Na2CO3
(aq) + HCl (aq)
à
Video:
http://www.youtube.com/watch?v=gtcE8TosEq4
Note for M-5H1:
Neutralization ion Exchange Reaction:
1. When an acid
reacts with a base, salt plus water are the products
Gas Forming Ion Exchange Reactions:
2. When either H2SO3;
H2CO3 or NH4OH is formed as a product it
immediately
decomposes thus demonstrating a gas forming
reaction.
3. Most books do not
show either H2CO3 or NH4OH as products, just
the
decomposed products of the
gases and water in the answer.
If H2CO3
is a predicted product in ion exchange, it is written as
CO2 and H2O.
For example:
Na2CO3
(aq) + HCl (aq)
à [H2CO3](aq)
+ NaCl (aq)
Should
be written:
Na2CO3
(aq) + HCl (aq)
à CO2(g) + H2O
(l) + NaCl (aq)
Two
other products which are shown differently:
[NH4OH] à NH3 + H2O
[H2SO3 ] à SO2 + H2O