CHM1032C
Module Homework Packet Name:___________________
Chapter 1i: Matter (Formerly Module 1)
A. _____(01) Matter Chart- Section 1.3 Corwin Fig 1.7 Answers
A1.____ (01) Matter Chart Applications Answers
B. _____(02) Element-Symbol- Section 1.4 Corwin Table 3.3 Answers Fig 3.4 Tabl 3.2
B1._____(02) Element Flash Cards (Hard Copy or Online)
B2._____(01) Element Identification Homework/Mixer (Click on Element for Answer)
C. ______(01) Element Classification- Section 1.5 Corwin Fig 3.5 Answer
C1.____ (01) Compounds and Chemical Formulas Section 3.3 Answers
D1,____ (01) Chemical and Physical Properties Section 1.7 Answers
D. _____(01) Chemical/Physical/Nuclear Change Sec 1.7 Answers Fig 3.1 Fig 3.6 Fig 3.11 Fig 3.12
E.
_____(01) Forms
of Energy Sec 1.13/7.1/lecture Answers Fig 3.13 Fig 3.14 Fig 3.15 Fig 3.16
E1.____ (01) Energy Transformations Lecture See Practice/Concept Exercise Answers
F. _____(03) Phase Diagram (From Lecture) Section 1.2 Answers
F1.____
(01) Phase Diagram Applications
_______(17)
Module 1i Total (First Exam)
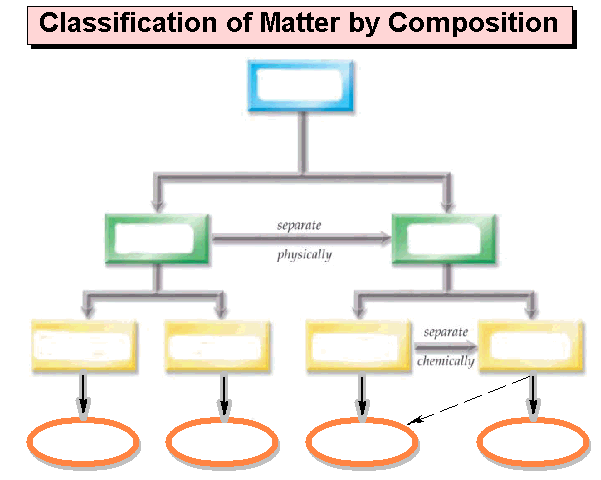
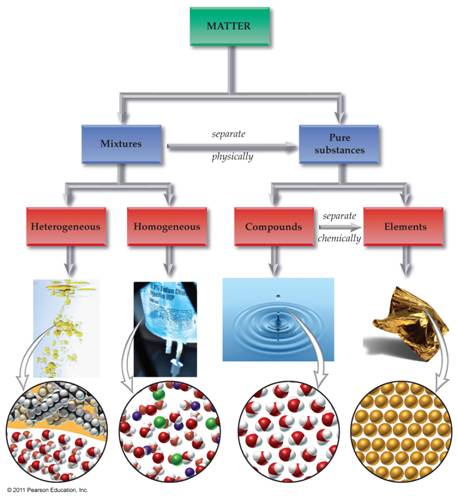
Chapter One: Part A Matter Chart 01
point
Draw below a matter
chart similar to the chart in section 3.1
page 47 of the Hein Foundations Chemistry
text or it may be of your own design as long as it clearly denotes lines which
describe which words are subunits of the more general word. The chart should include the following: homogeneous mixtures, heterogeneous
mixtures, Matter, Pure Substances, Mixtures, Compounds, Elements, Solutions,
Atoms, Molecules/Formula Units, and Colloids/Suspensions. Also draw/label the arrows: Separate
Physically and Separate Chemically:
Matter Chart Homework Critical Thinking:
Chapter
One: Part A1 Matter Chart-Critical Thinking Application 1
point
1. Where would you
place: colloids in the matter chart?
2. If you subdivided Inorganic Compounds and Organic
Compounds under compounds.,
sketch below where would you put: Salts, Acids, Bases, Covalent
Compounds?
3. Sketch below and show under which subdivision would you put:
Electrons, Protons, and Neutrons
4. . Sketch below and
show under which subdivision would you put:
electrons, protons,
neutrons, nucleus, orbitals
Chapter
One
Homework Packet
Hein 14th
Edition List
CHM 1032C Chapter One
Homework Packet
Chapter One Part
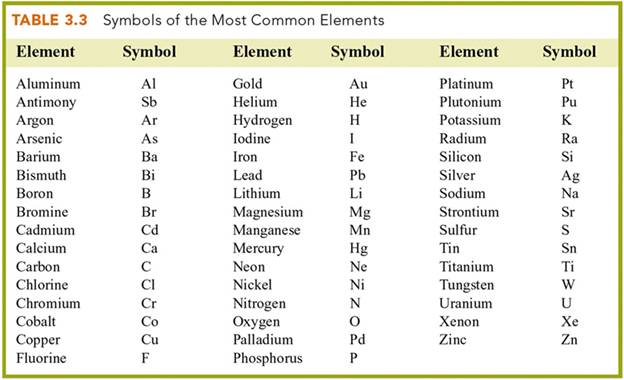
B: Element/Symbol 2 points
In the blanks below, write the symbol
for the element listed , or write the name (with
correct spelling for the element represented by its symbol:
1. Magnesium _____ 21. Cl __
___________
2. Manganese _____ 22. F _____________
3. Tungsten _____ 23. I _____________
4. Platinum _____ 24. Br _____________
5. Gold _____ 25. Zn _____________
6. Silver _____ 26. H _____________
7. Iron _____ 27. O _____________
8. Tin _____ 28. N _____________
9. Helium _____ 29. C _____________
10.
Antimony _____ 30. K _____________
11.
Lead _____ 31. P _____________
12.
Argon _____ 32. B _____________
13. Neon _____ 33. Al _____________
14. Krypton _____ 34. Cr _____________
15. Arsenic _____ 35. Ca _____________
16. Mercury _____ 36. Bi _____________
17. Copper _____ 37. Sr _____________
18. Cobalt _____ 38. Si _____________
19. Beryllium _____ 39. Ni _____________
20. Selenium _____ 40. Li _____________
Chapter One Part
B1: Element/Symbol
Paper or Online Flashcards 2 points
http://www.lsua.info/chem1001/elementquiz/elementquiz.html
CHM
1032C Chapter One: B2 Element Identification Homework
01point
Identify each of the following elements chalk board representation:


1. Element: _______________Symbol:_____ 2.Element: _______________Symbol:_____
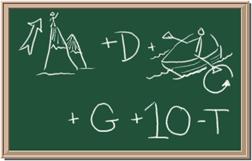
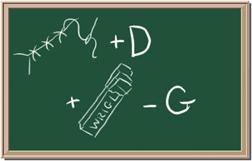
3. Element: _______________Symbol:_____ 4.Element: _______________Symbol:_____
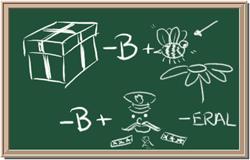

5. Element: _______________Symbol:_____ 6.Element: _______________Symbol:_____
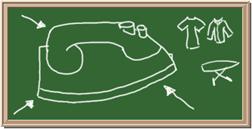
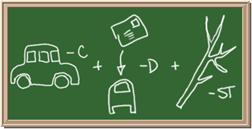
7. Element: _______________Symbol:_____ 8.Element: _______________Symbol:_____

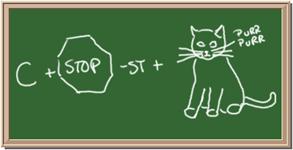
9. Element: _______________Symbol:_____ 10.Element: _______________Symbol:_____
CHM1032C Chapter One Homework
Packet
Chapter One Part C: Element
Classification 01 point
Using
the periodic chart provided, in the blanks below write the classification of
the elements listed:

The
classifications of the elements are:
Metals;
Semimetals
(Metalloids);
Nonmetals-Regular;
Nonmetals-Nobel
Gases;
Nonmetals-Class
by Itself
Use
the above classifications to classify each of the following elements.
You
must split the nonmetals into three subclasses:
1. 12C6 ______________
2. 39K19 ______________
3. 11B5 ______________
4. 1H1 _________________
5. 4He2
_________________
6. 56Fe26 _________________
7. 238U92 _________________
8. 73Ge32 _________________
9. 9Be4 _________________
10. 86Rn222 _________________
CHM1032C Module One Homework Packet
Part
C1: Compounds and Chemical Formulas (Section3.4) 2 points
1.
State the Law of
Definite Composition:
2.
Define Molecule:
3.
The chemical
formula for vitamin B3 is: C6H6N2O
In One Molecule How Many:
_____ carbon
atoms
_____ oxygen
atoms
_____ total
atoms
4.
Write the
chemical formula for Vitamin B6 which has eight Carbon atoms, 11
Hydrogen atoms, one Nitrogen atom, and three Oxygen atoms:
How many
total atoms are there in one Vitamin B6 molecule?
5.
Citric Acid (in
citrus fruit) has the following chemical formula:
C3H4OH(COOH)3
In one
molecule of Citric Acid, How many:
______Carbon
atoms
______Oxygen
Atoms
______
Hydrogen Atoms
______ total atoms
How
many atoms are in the following complex ion molecule:
[Cr(N2H4CO)6]4[Cr(CN)6]3 _______atoms
CHM1032C
Module One Homework Packet
Part D: Chemical/Physical/Nuclear Change 2 points
State whether each of the following is a physical change, a chemical change,
or a nuclear change:
__________________1. Electricity
decomposes water.
__________________ 2. Methanol
dissolves in gasoline
__________________ 3. Dry ice pellets
disappear
__________________ 4. Iron oxidizes to
rust
__________________ 5. Bromine vaporizes
into a reddish-brown gas
__________________ 6. Uranium-235
splits into two small elements when bombarded
with neutrons in an atomic bomb.
___________________7. Copper conducts
heat
___________________8. Baking soda
fizzes in vinegar
___________________9. Grinding sugar
crystals into a powder
__________________10. Sodium reacts
with chlorine gas
__________________11. Adding air to a
tire
__________________12. Slicing an orange
into wedges
__________________13. Hydrogen atoms
fuse into helium atoms in a hydrogen bomb
__________________14.” Dry ice”(Solid Carbon dioxide) vaporizes into a gas at room
temperature and sea level pressure
__________________15. Natural Gas burs with a blue flame
CHM1032C
Chapter One: Sample Exam Homework
Part D1: Chemical/Physical/Nuclear Properties (Section 3.6) 1 point
1. Define: Physical Property:
2. Chemical Property:
3. Classify each of the following as a chemical or physical property:
a.
Color
_________________
b.
Odor
_________________
c.
Reaction with water:
____________________
d.
Solubility in water: ____________________
e.
Melting point: ____________________
f.
Boiling point: ____________________
g.
Sublimation Point: ____________________
h.
Reaction with oxygen
____________________
i.
Density:
____________________
j.
Solid state:
___________________
k.
Reaction producing a Gas: _____________________
l.
Conductor of electricity: _____________________
m.
Water is insoluble in gasoline:
_____________________
n.
Good conductor of heat: _____________________
o.
Two chemical when mixed gives of heat:
___________________
p.
Appearance at Room Temperature: ___________________
q.
An element turns black when heated ___________________
r.
Silver tarnishes in Air ____________________
s.
An element is radioactive: ________________
CHM1025C Chapter One Homework
Packet
Part
E: Energy/Forms of Energy 1 point
Define Energy:
What is the difference
between the two types of energy:
potential and kinetic energy?
List the six different forms
of energy as an energy wheel as stated in the book:
Define the Law of
Conservation of Energy:
Reword the Law of
Conservation of Energy into the first law of thermodynamics:
Which state of matter has the
lowest kinetic energy?
What is the “Heat Death
Theory of the Universe”?
Bonus: define Gravitational
Energy; Tidal Energy; Sound Energy; Magnetic Energy; Radiant Energy; Dark
Energy
CHM1025C Chapter One Homework
Packet
Part
E1: Energy Transformations 1 point
Identify two forms of energy
that are involved in each of the following energy conversions:
1.
relating to a fossil fuel plant:
a. Burning coal converts water to steam:
b. A turbine spins and drives an
electrical generator:
2.
Solar Hot Water Energy
Panels:
3.
Photovoltaic
Solar Energy Panels:
4.
Radioactive
emissions vaporize water into steam
5.
Identify two
forms of energy that are involved in each of the following devices:
a.
Flashlight
b.
Solar Calculator
c.
Lead-acid battery
6.
Classify the
following energy sources as renewable and nonrenewable:
a. Biomass f. Hydropower
b. Petroleum g.
Coal
c. Geothermal h.
natural gas
d. Wind i. solar
e.
Uranium j. propane
CHM1025C Chapter One Homework
Packet
Part
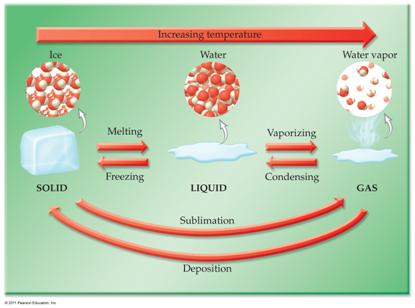
F: Phase Diagrams 3 points
Identify the
points labeled on the
Phase Diagram of water:
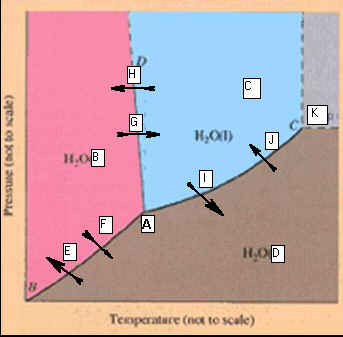
_____________________A.
_____________________B. _____________________H
_____________________C. _____________________I
_____________________D. _____________________J
_____________________E. _____________________K
_____________________F
_____________________G
CHM1025C Chapter One Homework
Packet
Phase Diagram for Carbon Dioxide.
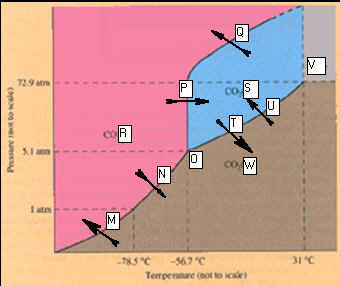
_____________________M.
____________________T
_____________________N. _____________________U
_____________________O. _____________________V
_____________________P. _____________________W
_____________________Q.
_____________________R
_____________________S
CHM1025C Chapter One Homework
Packet
Part
F1: Phase Diagram Applications
1 point

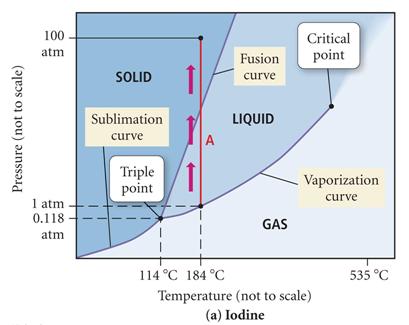
A demonstration of heating
iodine in a beaker has purple vapors..can you explain
using the phase diagram above?
Dr.
Lukacs Pod Cast Chapter 1 McMurry 7th
Registration:
http://weinstruct.org/weinstruct/main/course/1
Chemistry
Essentials
http://weinstruct.org/weinstruct/main/lecture/4
Chapter 1ii: ChemMath and Measurement (Hein 14th Chapter 2)
G. _____(01) Significant Figures- Section 1.9 Answers
H. _____(01) Round Off/Math of Significant Figures- Section 1.11 Answers
I. ______(01) Scientific Notation Section 1.10 Answers
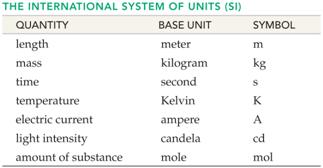
J. ______(01) Metric Basic Units /Numerical Prefixes- Section 1.8 Table 2.1 Answers
K. _____(01) Metric System Conversion Factors- Section 1,8 Answers
N. _____(05) Unit Analysis Sections 1.12 Answers Pretest #2 Ans2 Online Site
O. _____(02) Temperature Conversion Section 1.13 Answers
P.
______(02) Density/Specific
Gravity/Volume Calculations Section 1.14 Answers hi
Q. _____ (02) Specific Heat Calculation/Ice Cube Problem Section 1.13 Answers hi
_______(16)
Chapter 1ii Total (First Exam)
Chapter One-Part G: Significant figures 1 point
In the
blank, state the number of significant figures in each of the following
measurements:
____1. 0.05 mL
____2. 250.0 cm
____3. 456,000,000 people
_____4. 1000 g
_____5. 0.00006500 moles
_____6.
0.00200 kg
_____7.
50 seconds
_____8.
50.0 Seconds
_____9.
50.00 Seconds
_____10.
0.05 Seconds
Significant Digit Animation:
http://www.lsua.info/chem1001/Chap2-3Movies/sigdigit.html
Chapter One Homework Packet
Chapter 1-Part H: Rounding Off & Arithmetic
Operations of Sign. Figures 1 point
Round
off the following numbers to three significant figures:
(1)
1.598 x 106 = _____________
(2)
0.000 000 484 500 = _________________
(3) 0.01045 =
_______________
(4) 1.98754 X10-7 = ________________
Perform the following addition/subtraction/multiplication/division
operations and express the answer using the proper units and significant
figures:
(5) 4
mL
16.3
mL
+ 0.953 mL
(6) 376.5 mL
- 76
mL
(7)
16.5 cm
X
1.7 cm
(8) 12.0 g ÷ 1.00 g =
or
12.0 g
/ 1.00 g =
(9)
9.2 cm X 9.20 cm
X 3.14 X 22.65cm
=
(10)
(5398 cm3 – 2060.2 cm3) /16.8 cm3/sphere =
CHM 1032C Chapter One Homework Packet
Chapter One-Part I: Exponential Numbers and Scientific
Notation 1 point
Express
the following ordinary numbers in scientific notation (If greater than three
significant figures, round off to three significant figures:
(1)
1,010,100,000,000, 000
= ________________
(2)
0.000 000 000 000 019 = ________________
(3) 456,789
= _________________
(4)
0.0001198 = _____________
(5)
1,000,000 = ______________
(6) 0.000200 =
______________
(7)
Express the following
products in exponential form
2 X 2
X 2 X 2 X 2 X 2 X 2 X 2 = ______________
(8)
and use your calculator to calculate the value:
Value
= ___________________
(9) Express the following powers often notation:
1 x 100
= ______ 1 X 101=______ 1 x 10-1 = _________
(10)
Express the ordinary number in scientific notation in three significant
figures:
60,230,000,000,000,000,000,000
= _______________________
Chapter One Homework Packet
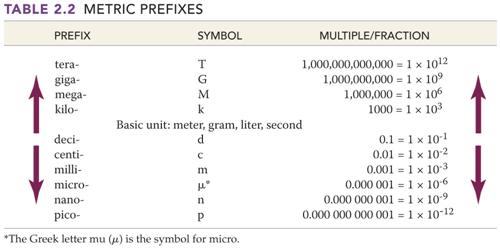
Part J: Metric
System Basic Units/Numerical Prefixes
1 point
Fill in the blank with the proper basic
unit or metric prefix, then in the parenthesis put the unit’s or prefix’s
abbreviation (Use table from Chapter 3):
____________( )
1. Basic unit of length in the metric system
____________( )
2. Basic unit of volume in the metric system
____________( )
3. Basic unit of mass in the
metric system (not SI)
__________( )
4. Metric prefix which means 1/1000 of a unit
__________( )
5. Metric prefix which means 1000 units
__________( )
6. Metric prefix which means 1/100 of a unit
__________( )
7. Metric prefix which means 1/10
of a unit
__________( )
8. Metric prefix which means
1,000,000 units
__________( )
9. Metric prefix which means
1/1000000 ( 10-6)
of a unit
__________( ) 10.
Metric Prefix which means 1/1000000000 (
10-9) of a unit
Metric Prefix Table:
http://www.lsua.info/MetricSystem/MetricPrefix.html
Metric System Animation:
http://www.lsua.info/chem1001/Chap2-3Movies/metric.html
CHM 1032C Chapter One Homework Packet
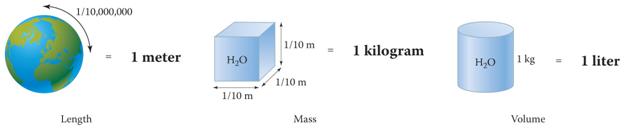
Chapter One-Part J
Metric Unit Factors 1 point
Fill in the blank with the number which
completes the metric unit factor:
(1)
__________mg = 1.000 g
(2)
__________mg = 1.000 kg
(3)
__________mL = 1.000 L
(4)
__________cm = 1.000 m
(5)
___________mL = 1.00 cm3
(6)
____________km = 1.000 m
(7) ____________ g =
1 kg
(8) ____________ cm = 1 dm
(9) ___________ µL
= 1 L
(10) __________ nm = 1 m
(11)
Write a unit equation for each of the following metric equivalents:
(a) M and Tm (b) L and mL (c) Bytes and G-bytes
(a) ______________ (b) ____________ (c)
________________
|
|
|
Chapter One Homework Packet
Part N: Unit
Analysis Problems 5 points
(Work any 10-you must show dimensional
analysis sequence for credit)
Apply the unit analysis method of
problem solving to each of the following
( If greater than three significant figures round off to three
significant figures):
Problem 1
An
oxygen molecule travels 975 mi/hr at room temperature. There are 5280 ft = 1
mi; 12 in = 1 ft, 2.54 cm = 1 in, 1.6 km = 1 mi, and 3600 sec = 1 hr. What is
the velocity in meters per second?
Problem 2
If one gram is equal to 15.4 grains. How many 5.00 grain
aspirin tablets may be made from 1.00 kilogram of aspirin?
Problem 3
A
parsec is the distance light travels in 3.26 years. Given the velocity of
light, 3.00 x 108 m/sec, how many kilometers does light travel in
one parsec?
Problem 4
I have
1400 radio programs I want to put on an Apple Ipod. Each program requires 5
megabytes of disk space. If there are 1024 megabytes in a
gigabyte. How many gigabytes of disk space do I need minimum to store
all my programs on the IPod. The Mini-Ipod holds only 4 gigabytes of recordings, could I use a mini for my project?
Problem5
Find the mass in grains of a 325 milligram aspirin tablet.
(Given: 1.00 g = 15.4 grains)
Problem6
Insurance statistics state that a person loses 8 minutes of average life for
each cigarette smoked. If there are 20 cigarettes in a pack and the average
cost of cigarette is $5.00 per pack over the next 25 years, how many years of
average life would a person lose for smoking 1.5 packs a day for 25 years?
Problem7
What is the density of water in lb/ft3, if the density of water at
25oC is 1.00 g/ml?
[Hint: There are 2.54 cm = 1 in (or 16.48 cm3
= 1 in3); 454 g = 1 lb ]
Problem8
Calculate the velocity of a car traveling car traveling 65 miles/hr in ft/sec.
Problem9
How many milligrams does a 0.750 carat diamond weigh?
(Hint: 1 carat = 0.200 g)
Problem10
Diamond has a density of 3.513 g/cm3. The mass of a diamond is often
measured in carats, 1 carat equaling 0.200 g. What is the volume of a 1.50
carat diamond?
Problem11
Liquor used to be sold in fifths. A fifth is one fifth of a gallon. A gallon is
128 fluid ounces. Today liquor is sold in bottle sizes of 750 ml to equate to
the old fifth. If there are 946 ml in a quart, calculate the number of
milliliters in a fifth. How many milliliters difference is there in the
bottling?
Problem 12
1. On
July 23, 1983 Air Canada Flight 143, flying at 26,000 feet from Montreal to
Edmonton, ran out of fuel because the first officer ask
the mechanic for the conversion factor of mass to volume at Montreal. The
mechanic gave the first officer the answer 1.77 with no units. The plane had
7682 L of fuel at Montreal. The pilot knew he needed 22,300 kg of fuel to make
the trip. The mechanic's answer of 1.77 was pounds per liter not kilograms per
liter caused the error such that only 4917 L of fuel was added. If there are 2.205
pounds in a kilogram, how many liters of fuel were needed for the trip? How
many liters minimum of fuel should have been added at Montreal before takeoff?
Problem 13
Before
1982 the US Mint cast penny coins from an alloy of copper and zinc. A 1980
Penny weighs 3.051 g and contains 2.898 g of pure copper. In 1982 the US Mint
stopped making copper pennies, because the price of copper was worth more than
the penny. The post 1982 penny contains only a layer of copper over zinc. A
1990 penny weighs 2.554 g and contains 2.490 g of zinc. If the mint melted down
one pound of 1980 pennies, how many 1990 pennies can be made from the total
copper from the 1980 pennies?
Problems 14
An
Olympic size swimming pool is 50.0 m long and 25.0 m wide. How many gallons of
water ( d = 1.0g/mL )are needed to fill the pool to an
average depth of 5.5 feet.
Problem 15
A
furniture factory needs 29.5 ft2 of fabric to upholster one chair. A
Europen supplier sends the fabric in bolts of exactly 200 m2. What
is the maximum number of chairs that can be upholstered by three bolts of fabric. Hint: 1 m - 3.281 ft)?
Problem 16
My throw
away car gets 23.4 mi/gal and hold 70.1 L of gasoline. How far can I drive on a
tankful of gas?
If gas
cost $3.49/gal; how much does a tankful of gas cost?
If the
average speed on a trip is 92.2 km/hr, How many hours
may I drive the car on the trip before I run out of gas?
CHM 1032C Chapter One Packet
Part O Temperature
Conversion 2 points
The general formula
for the conversion of temperatures on scale X to temperatures on scale Y is:
oY = Y units/ X units ( oX - RPx) + RPy
1.Write
the formula for the conversion of Fahrenheit to Celsius:
2. Write the formula for the conversion
of Kelvin to Celsius:
3. convert
-196oC to oF
4. convert
-196oC to K
5. The Rankin scale uses a Fahrenheit
unit, but assumes zero to be absolute zero.
If absolute zero on the Kelvin scale is zero and on Celsius scale is
-273oC, calculate
absolute zero on the Fahrenheit scale, then estimate the Freezing point of
water on the Rankin scale. (The BP water=212
oF=100 oC=373K)
CHM 1032C Chapter One Homework Packet
Chapter One-Part P: Density, Specific Gravity & Volume
Problems 2 points
1. A
quartz rock was cut into a rectangular solid paperweight. IF the paperweight has a mass of 165 g and
measures 5.00 cm by 5.00 cm by 25.0 mm, what is its volume in cubic
centimeters?
2. Calculate the density in g/mL for 10.0 grams of ethyl ether having a volume
14.0 mL.
Part Q: Heat and
Specific Heat 2 points
- Find the specific heat of gold if 25.0 cal is required to heat
30.0 g of gold from 27.7 oC to 54.9 oC.
- Calculate the heat required to raise to raise 25.0 g of iron (sp
Heat=0.108 cal/g ∙ oC) from 25.0 oC to 50.0 oC.