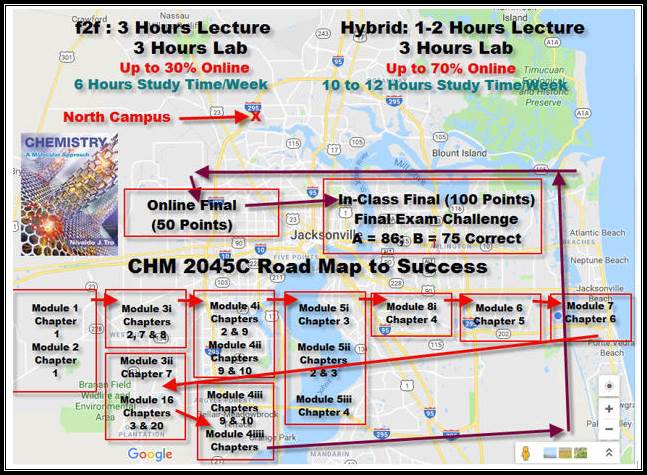
CHM 2045C
Road Map Spring 2018
ON-Line Grade
Calculator: http://www.fccj.us/chm2045/45grdcal.html
Exam Outline based on 4th Edition of Tro’s Text:
Module One: Matter, Measurement and Problem Solving
(Chapter
1) ppt
A. _____(10) Matter Chart-Section 1.3 Answers
A1._____(05)
Critical
Thinking Matter Chart Apps Answers
B. _____(20) Element-Symbol-Section 1.3 Table xxx Answers Element List
C.
_____ (05) Element
Classification-Section
1.3 Answers ck
C1.____
(10) Compounds
and Chemical Formulas Section
3.3 Answers
F.
_____ (15) Phase
Diagram
Section 1.3; 11.8 Answers Real Video Movie
_______(65) Total
M-1 Required/Suggested Homework:
B1. ______(05) Online/Flash Cards Element Homework
______(00) M-1 Hardcopy Homework/Study Packet (Suggested)
_____(30) Safety
Lab (Week 1)
______(30)
Scientific Method Project: The Controlled Experiment (Lab) (Week 2)
Module Two: ChemMath and Measurement (Chapter 1)
A. _____(05) Significant
Figures-Section 1.7 Answers
B. _____(05) Round Off/Math
of Significant Figures- Section 1.7 Answers
C. _____(05) Scientific
Notation Section 1.7 Answers
D. _____(05) Metric Numerical
Prefixes T1.5-Section 1.4 Answers
D1. ____(05) Metric Basic Section
1.6 . Answers
D2.____ (05) Derived Units:
Volume and Measurement Section 1.5 T1.6 Answers
E. _____(05) Metric System
Conversion Factors-Sections 1.6 Answer
F. _____(20) Unit Analysis
Problems Section 1.8 Answers Pretest #2 Ans2
Online Site
G. _____(10) Temperature
Conversion Section 1.6 Answers
H. _____(10) Density/Specific
Gravity & Volume Calculations Section 1.6 Answers
_______(75) Total
M-2 Required/Suggested Homework:
______(00) M-2 Hardcopy
Homework/Study Packet)(Suggested)
_____ (60) Gasoline Project (Submit
last week of class) Sample Table (2
Labs)
_______ (30) Temperature Project (Lab)
(Week 2)
_______ (30) Measurement Lab (Week 3)
_______ (30) Density/Volume Lab
(Week 4)
_______ (30) Specific Heat/Calorimeter Lab (Week 4)
Jespersen
7th cross referenced Tro 4th
Through M-1 and M-2
Module Three Part I: Atomic
Theory & The Periodic Chart (Chapter
2, 5,
6)
Assignment
Outline
A. _____ (05) Atomic Notation-Section
2.6 Answers ppt2 ppt5 ppt6
B. _____ (32) Electron Configuration-Sections
7.6, 8.3, 8.4 Answers
C. _____ (10) Orbitals/Subshells of the
Periodic Table-Section 8.4 Answer
C1.
____ (18) Spectroscopic
Notation from Periodic Chart Section 8.4 Answer
D. _____ (05) Electron Dot Structures-Section
9.3 Answers
E. _____ (05) e-1
Configuration of Ions-Section 8.7 Answers
F. _____ (05) Periodic Ionic
Character-Section 2.7 Answers
P. ____ (10) Periodic Table
Properties Chapters 2, 7, 8 Answers
_______(90) Total
Jespersen
7th cross referenced to Tro 4th
M-3
M-3 Required/Suggested Homework:
______(00) M-3 Hardcopy Homework/Study Packet (Suggested)
Jespersen 7th is not cross
referenced with Tro 4th for the following:
(Values
may be changed)
Module Four Part I: Language of Chemistry/Chemical Bonds(Chapter2,6,7) Assignment Outline
A. _____(10) Bond Recognition/Compound
Classification-Sections 2.8 Answers
B. _____(40) Dot Structures of Molecules-Section
8.7 Answers
C. _____(05) Binary Molecular(Covalent)
Compounds-Section 2.8 Answers
D
_____(05) Binary
Ionic Compounds-Section 2.5-2.6 Answers
E. _____(20) Polyatomic Ions-Section
2.5-2.6 Answers e
F. _____(20) Ternary Ionic Compounds-Section
2.5-2.6 Answers f
G. _____(05) Binary Acids/ Ternary
Oxyacids-lecture, 4.5
Answers g
H. _____(25) Inorganic Compounds
Section 2.8 p97-98 Answers h
_______(130) Total = ______%
Jespersen
7th cross referenced to McMurry 6th M-4i
M-4 Required/Suggested Homework/Study
Packet:
______(00) M-4 Hardcopy
Homework/Study Packet
______(105)
Online Names/Formulas Homework (Submit
separate Goldenrod form on M-4 Exam Day)
______(10) Polyatomic Ions Flash Card or Progressive
Polyatomic ion online Homework
______(60) Polyatomic Ions Progressive Test (Best Score of
three attempts) Required List
_____ (30) Hard Copy Dot Structure Homework/Lab:
Module Five Part I:Chemical
Equations & Stoichiometry (Chap
3 & 4)
Module 5ii
A._____(05) Molecular Mass
Calculation-Section 3.3 Answers a ppt3 ppt4
B._____(05) Mole Calculations-Section
3.3 Answers bcd
C._____(05) Percentage Composition
Calculation-Section 3.10 Answers bcd
D._____(05) Empirical Formula
Calc. from % Comp & Analysis-Section 3.11 Answers bcd
Nodule 5i
E._____(10) Symbols Chemical
Reactions-Chapter 3 see sample Answers ef
F._____(10) Balancing Chemical
Equations by Inspection-Section 3.1 Answers ef
G._____(10) Predicting Single
Replacement Products . Section 4.8 Answers g
H._____(10) Predict Double
Replacement & Organic Combustion Products 4.1, 4.3, 4.5 Answers h
Module 5iii
I.______(10) Mole-Mole Problems
Section 3.3 Lecture Answers ij
J._____ (10)
Mass-Mass
Stoichiometric Problems-Section 3.3 Answers ij
K._____(10) Excess/Limiting Reagent
Problems-Section 3.5 Answers kl
L._____(10) Impure reagents/% Yield
Problem-Section 3.4 Answers kl
N._____(10) Multi step Synthesis
Problems-Lecture Answers
O._____(10) Mixture Problem
Lecture Answers 2nd
Example Combined Example
P. ____(10) Empirical Formula
from Analysis – Answers Section 3.11
______(145) Total = ______%
M-5 Required/Suggested Homework:
_____ (00) M-5 Hardcopy Homework/Study Packet
______(??)
M-5ii Multiple Choice (MC) Practice (Blackboard Online)
Module Eight:
Solutions/Oxidation & Reduction (Chapter
4) ppt
Assignment Outline
A. _____(05) Solution Properties-From
Lecture Answers
B. _____(05) Factors Affecting Rate
of Dissolving-From Lecture(11.4) Answers
C. _____(05) Writing Ionzation Reactions acids/salts Sect 4.2, 4.3 Answers
D. _____(10) Solution Preparation
Problems-Section 3.6, 3.7 Answers
E. _____(10) Solution Dilution
Problems-Section 3.7 Answers
F. _____(20) Solution Reaction
Problems-Section 3.9-3.10, 4.10 Answers
G. _____(20) Rewrite Equations
Ionically –Section 4.3 Answers
H. _____(30) Redox
Equations-Sections-Section 4.6, 4.7, 4.9, 4.10
Answers
K. ____
(10) Solution
Definitions-Sect 3.6, 11.3, From Lecture Answers
L. _____(10) pH calculations-Section
14.5, 14.6 Answers
______(120)
Total = ______%
M-8 Required/Suggested Homework/Study:
______(00) M-8
Pretest Hardcopy Homework/Study
Packet
G2.
______(20) Hard Copy (Acid Media) Writing Net Ionic Reactions Homework
(Sect 14.11)
H1. ______(30) Hardcopy REDOX Homework Pre/PostLab
Required) (15 equations)
H2. ______(48)
12 rounds in Lab
Module Seven:
Thermochemistry & States of Matter (Chapter
8) ppt
Assignment Outline (Kotz)
A. ____(05) First Law of
Thermodynamics & Related Terms-Sect 8.1-8.3, 8.13-8.14 Answer
B. ____(10) Discussion Questions-Chapter
8 Answer
C. ____(10) Specific Heat Problem-Section
8.8 p287-8 Answer Sample 2
D. ____(10) Enthalpy Change with
Phase Change Prob-Sect 8.7 Answer Sample 2
E. ____(05) Enthalpy Change in
Chemical Reaction-Section 8.6, 8.12 Answer
F. ____ (05) Bomb Calorimeter
Section 8.7 Answer
G. ____(20) Hess Law
/Heats of Reaction Problem-Section 8.8 Answer
H. ____(10) Standard
Enthalpies of Formation Problem-Section 8.9 Answer
J. ____ (10)
Introduction to Entropy and Spontaneity-Section 8.12, 8.13
L. ____ (10)
Introduction to Free Energy and Spontaneity-Section 8.13
______(115)
Total = ______%
M-7 Required/Suggest Homework/Study:
______(00) M-7 Pretest Hardcopy
Homework/Study Packet
______(50) M-7
Multiple Choice (MC) Practice (Blackboard Online)
Module Six: The Gaseous State (Chapter 9) Assignment Outline (Kotz)
A._____(05) Kinetic Molecular
Theory-Section 9.6 Answer ppt9
B._____(10) Discussion Real vs
Ideal Gas Equation-Sect 9.8 Answer bc
C._____(05) Standard
Conditions/Molar Volume-Sect 9.1-9.2 Answer bc
C1._____(10)
Gas pressure Calculation/Value of R Calculation
D._____(15) Gas Laws/Vocabulary-Sections
9.2-9.3, 9.5, 9.7 Answers
E._____(30) Gas Law Problems-Sections
9.2-9.3, 9.5, 9.7 Answers
F. _____(05) Volume-Volume
Stoichiometry Problem-Section 9.4 Answers fg
G._____(10) Mass-Volume
Stoichiometry Problem-Section 9.4 Answers fg
H._____(10) Gas
Densities/Molecular Mass Determination-Sect 9.4 Answers
______(120)
Total = ______%
M-6 Required/Suggested Homework/Study:
______(00) M-6
Pretest Hardcopy Homework/Study
Packet (gray)
Cross
Reference from 5th to 6th edition stops here, below 5th
edition
Module Three Part III:
Atomic Theory & The Periodic Chart (Chapter
7)
G: _____(10) Frequency from Wavelength Calculation Section 7.1
H. _____(10) Balmer-Rydberg Equation Calculation Section 7.3
I. _____(10) De
Broglie Wavelength Calculation Section 5.5
Q. _____ (20) Quantum Numbers-Section
7.5-7.6 Answers
S. ____
(10) Orbital Shapes/Nodal Surfaces-Section 7.9
K1____
(10) Vocabulary Chapter 7 p 344-345
N. ____
(10) Discussion Questions-Chapter 7 (additional p346-348)
_______(80) Total =
______%
Jespersen 7th cross
referenced to McMurry 6th Module 3ii only above
Module Four Part III: Chemical Bonding & Molecular
Structure (Chapters 8-9)
L. ____ (10) Bond Angles/Bond
Lengths Steric#s 2-3-4 Section 9.2 Answers
L1. ____ ((10) Bond
Angles/Bond Lengths Steric#s 5&6-Section 9.2
M._____ (10) Molecular Orbitals Section 9.7-9.8
N. ____ (10) Geometry of Molecules-
Steric#s 2-3-4 Section 9.1-9.2 Answers
N1. ____
(10) Geometry of Molecules-Steric#s
5&6 Section 9.1-9.2
O. ____ (10) Polarity of Molecules-
Steri #s 2-3-4 Section 8.6, 9.3 Answers
O1. ____
(10) Polarity of Molecules- Steric#s
5&6 Section 8.6, 9.3
P. ____ (10) Hybrid Orbital
Recognition Steric#s 2-3-4-Sect 9.5-6 Answers
P1. ____ (10) Hybrid Orbital
Recognition Steric#s 5&6-Sect 9.5-6 Answers
P2._____(05) Valence Bond
Theory Section 9.4
Q. _____ (10) Formal Charge-Section 8.7 page 377-384
R. _____ (10) Resonance Structures Section 8.8
______(xx) Total = ______%
Jespersen 7th cross
referenced to McMurry 6th Module 4iii only above
ACS Toledo Chemistry Placement PreTest
Correct/Total Week One:
Part I General Mathematics Items 1-20 ____ / (20)
Part II General Chemical Knowledge Items 21-40 ____ / (20)*
Part III Specific Chemical Knowledge Items 41-60 ____ / (20)*
Math: ______(20) Chemistry:
_____ (40) Grand Total _____(60)
OR *Makeup Week Two
ACS California Placement PreTest
Correct/ Wrong
General Chemistry Knowledge only Items
1-44 ____ /(40)
Online Final Exam : _____(50) (100 questions)
Comprehensive Final Exam : _____(100)
(100 questions)
Final
Exam Challenge Grade: ____
(86-100%)
= A; (75-85%) = B (65-74%) = C (50-64%) = D