CHM
2046C Module-4ii Homework Packet Name: ____________________
Please
complete the following homework packet before you actually attempt the pretest
before or after class each day. This is homework, not the pretest. You attempt
it before you take the exam. You grade it. This completed packet is due the day
of the exam.
B._____ (05) Lewis Dot/Stick Structure via Octet Rule Steric#s 2-3-4 Sect 8.3/8.7Answer
E._____ (01)
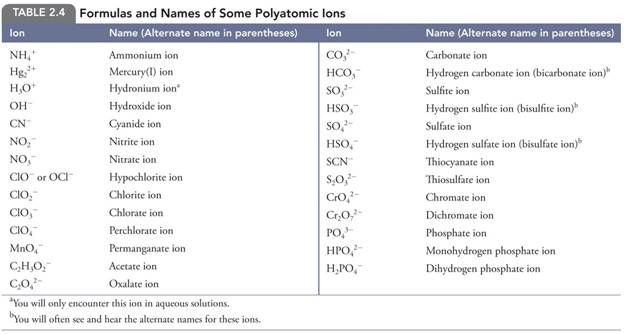
Polyatomic Ions Table
2.4 Page 85
L. ____ (01)
Bond
Angles/Bond Lengths Steric#s 2-3-4
Section 9.2 Answers
N. ____ (01) Geometry of Molecules- Steric#s 2-3-4 Section 9.1-9.2 Answers
O. ____ (01) Polarity of Molecules- Steri #s 2-3-4 Section 8.6, 9.3 Answers
P. ____ (01) Hybrid Orbital Recognition Steric#s 2-3-4-Sect 9.5-6 Answers
______(10) Total
Module
Four II: Part B Dot Structures of Molecules Review 05 points
Choose
Two from each question. Use the simple octet rule (but
you may use formal charge method for a more accurate drawing-Required in M4iii
B1). Using
a periodic chart/octetrule draw the electron dot/stick structures of the
following molecules:
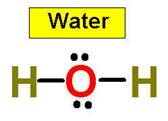
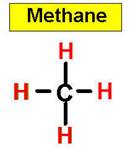
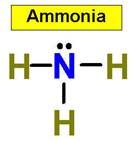
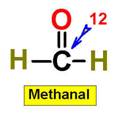
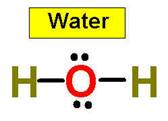
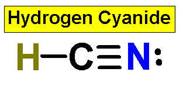
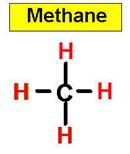
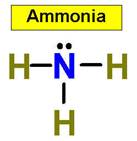
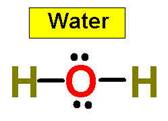
1. NH3 CH4 H2O2 H2O HCl
H2 O2 N2
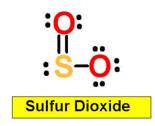
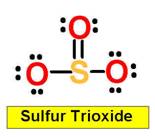
2. H2SO4 H2SO3 H3PO4
H3PO3 HClO4 HClO3 HClO2 HClO
3. HNO3 H2CO3 HNO2
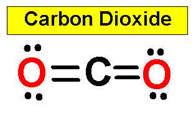
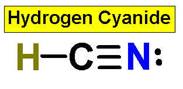
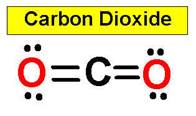
4. CO2 HCN
SiO2 SO3
5. HC2H3O2 H2C2O4 HCHO2
carbon to carbon by single covalent bond
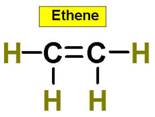
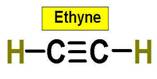
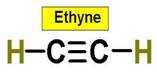
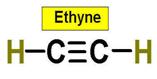
6. C2H4 C2H2 C3H8 C2H6
bond carbons to carbon
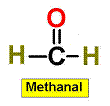
7. CH2O CH3COCH3
carbon
to carbons by single covalent bonds above
8. CH3OCH3 CHONH2
oxygen
separates O & N both
the carbons bond to C
9. CH2NH2COOH CH3CHNH2COOH
carbon to
carbons by single covalent bonds -NH2
hooks to #2 carbon in both above
10. CH3COOCH2CH3 CHOOCH3
carbon to
carbons by single covalent bonds (-CH3 also hooks to an oxygen
above)
(-CH2CH3 also
hooks to an oxygen above)
Reading
Reference: Sections 8.5 Octet Rule and 8.7 Formal Charge (page 374)
Reference:
B. Dot Structures of Covalent Compounds Section
2.10, 6.6, 7.1, 7.5, 7.6
Answers: http://www.fccj.us/chm2045/SampleTest/45M4bAnswers.htm
Required
List of Polyatomic Ions 0 Points
|
1 |
IA |
IIA |
|
|
|
|
H |
|
|
|
|
|
IIIa |
IVA |
VA |
VIA |
VIIA |
He |
|
2 |
|
|
|
|
|
|
|
|
|
|
|
|
B |
C* |
N |
|
|
Ne |
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Si |
P |
S |
Cl |
Ar |
|
4 |
|
|
|
|
V |
Cr |
Mn |
Fe |
|
|
|
|
|
|
As |
Se |
Br |
Kr |
|
5 |
|
|
|
|
|
Mo |
|
|
|
|
|
|
|
Sn |
Sb |
Te |
I |
Xe |
|
6 |
|
|
|
|
|
W |
|
|
|
|
|
|
|
Pb |
|
|
|
Rn |
|
7 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
Acetate ___________ Ammonium ___________ Antimonate ___________ Antimonite ___________ Arsenate ___________ Arsenite ___________ Azide ___________ Bicarbonate ___________ Borate ___________ Bromate ___________ Bromite ___________ Carbide __________ Carbonate __________ Carbonite __________ Chlorate __________ Chlorite __________ Chromate __________ Cyanate _________ Cyanide _________ Dichromate __________ Dihydrogen Arsenate
__________ Dinhydrogen
Phophate __________ Dihydrogen Phosphite
__________ Ferrate
___________ Hydrogen
carbonate ___________ Hydrogen
arsenate ___________ Hydrogen
phosphate ___________ Hydrogen
phosphite __________ Hydrogen
sulfate __________ Hydrogen
sulfite __________ Hydronium __________ Hydroxide ___________ |
Hypobromite __________ Hypochlorite __________ Hypoiodite __________ Hypophosphite __________ Hyposulfite ___________ Periodate __________ Iodate __________ Iodite __________ Mercury (I) __________ Manganate __________ Molybdate _________ Nitrate __________ Nitrite __________ Oxalate __________ Perbromate __________ Perchlorate __________ Permanganate __________ Phosphate __________ Phosphite __________ Plumbate __________ Plumbite __________ Selenate ___________ Selenite ___________ Silicate ___________ Stannate ___________ Stannite ___________ Sulfate ___________ Sulfite ___________ Tellurate ___________ Thiocyanate ___________ Tungstate ___________ Vanadate ___________ |
|
Reference Online Homework: Polyatomic Ions-Section 2.12 Answers e
http://www.northcampus.net/Nomenclature/PolyatomicIonFormula/ProgressivePolyatomicIonFormula.html
Required List of Polyatomic Ions
|
1 |
IA |
IIA |
|
|
|
|
H |
|
|
|
|
|
IIIa |
IVA |
VA |
VIA |
VIIA |
He |
|
2 |
|
|
|
|
|
|
|
|
|
|
|
|
B |
C* |
N |
|
|
Ne |
|
3 |
|
|
|
|
|
|
|
|
|
|
|
|
|
Si |
P |
S |
Cl |
Ar |
|
4 |
|
|
|
|
V |
Cr |
Mn |
Fe |
|
|
|
|
|
|
As |
Se |
Br |
Kr |
|
5 |
|
|
|
|
|
Mo |
|
|
|
|
|
|
|
Sn |
Sb |
Te |
I |
Xe |
|
6 |
|
|
|
|
|
W |
|
|
|
|
|
|
|
Pb |
|
|
|
Rn |
|
7 |
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
|
The following Polyatomic Ions
should be in you chemistry vocabulary:
|
Acetate CH3COO−
or C2H3O2− Ammonium NH4+ Antimonate SbO43− Antimonite SbO33− Arsenate AsO43− Arsenite AsO33− Bicarbonate HCO3− Borate BO33− Bromate BrO3− Bromite BrO2− Carbide
C22− Carbonate CO32− Carbonite
CO22− Chlorate ClO3− Chlorite
ClO2− Chromate CrO42− Cyanate OCN− Cyanide CN− Dichromate
Cr2O72− |
Dihydrogen
Arsenate H2AsO4− Dinhydrogen
Phophate H2PO4− Dihydrogen
Phosphite H2PO3 Hydrogen
carbonate HCO3− Hydrogen
arsenate HAsO42− Hydrogen
phosphate HPO42− Hydrogen
phosphite HPO32− Hydrogen
sulfate HSO4− Hydrogen
sulfite HSO3− Hydronium H3O+ Hydroxide OH− Hypobromite BrO− Hypochlorite ClO− Hypoiodite IO− Hypophosphite PO23− Hyposulfite SO22− |
Iodate IO3− Iodite IO2− Manganate MnO42- Mercury (I)
Hg22+ Nitrate NO3− Nitrite NO2− Perbromate
BrO4− Perchlorate
ClO4− Periodate
IO4− Permanganate MnO4− Phosphate PO43− Phosphite PO33− Plumbate PbO32− Selenate SeO42− Selenite
SeO32− Silicate SiO32− Stanate SnO32− Sulfate SO42− Sulfite
SO32− Tungstate WO42− |
Bonus 1 point Each: Using a current CRC Handbook of Chemistry/Physics find a polyatomic ion list in an inorganic compound that is not on the list below:
The Complete Polyatomic Ions List http://www.fccj.us/PolyatomicIons/CompletePolyatomicIonList.htm
Taylor’s ¾ Rule: http://www.fccj.us/PolyatomicIons/Taylor34OxygenRuleHandout.htm
Taylor’s Charge Rule: http://www.fccj.us/PolyatomicIons/TaylorChargeRuleHandout.htm
National Publication 2YC3 Newsletter (2014 vol iii)
http://www.2yc3.org/Newsletters/2014/2014iii.pdf
Jespersen’s Common Polyatomic Ions
CHM 2046C Progressive
Polyatomic Ions Jespersen 1
Point
Name:_____________________
Write the formula and the charge for the following
polyatomic ions: Jespersen(Table 2.4
page 85)
|
Name |
Formula with charge |
Name |
Formula with charge |
|
Ammonium |
|
Sulfite |
|
|
Mercury(I) |
|
Hydrogen Sulfite |
|
|
Hydronium |
|
Sulfate |
|
|
Cyanide |
|
Hydrogen Sulfate |
|
|
Nitrite |
|
Thiocyanate |
|
|
Nitrate |
|
Thiosulfate |
|
|
Hypochlorite |
|
Chromate |
|
|
Chlorite |
|
Dichromate |
|
|
Chlorate |
|
Phosphate |
|
|
Perchlorate |
|
Monohydrogen Phosphate |
|
|
Permanganate |
|
Dihydrogen Phosphate |
|
|
Acetate |
|
Hydrogen
Carbonate |
|
|
Oxalate |
|
Carbonate |
|
Module
Four II: Part L Bond Angles 1 point
What
is the bond Angle in the following structures:
___1. 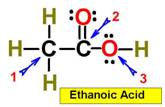 ___4.
___4.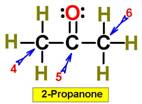
___2. ___5.
___3. ___6.
___7. 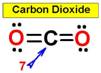 ___8.
___8. 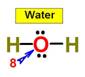 ____9.
____9.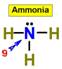
___10. 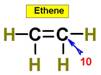 ____11.
____11. 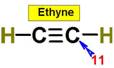
___12. 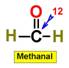 ___13.
___13. 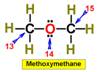 ___16.
___16.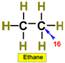
___14.
___15.
___17.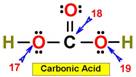 ___20.
___20.
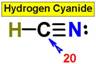
____18. _____19.
Bonus:
___21. 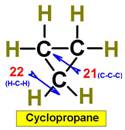 ___23.
___23. 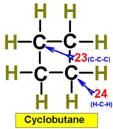
___22. ___24.
Steric
Numbers do not predict bond angles within rings of carbons
Reading
Reference: Jespersen 7th Section 9.2;
Work
End of Chapter Problems #9.83-9.84 page 461
Module Four II - Part N: Geometry of Molecules 1 point
Use the dot/stick structures on the Part L page to state the geometry of the molecules:
Steric # = 2, 3,or 4:
Bent Linear Trigonal Planer Planer Trigonal Pyramidal Tetrahedral
Steric # = 5 or 6
Trigonal-bipyramidal Square Planer Seesaw T-shaped Octahedral
|
_____________1.
H2O _____________2.
CO2 _____________3.
C2H4 _____________4.
SO2 _____________5.
SO3 _____________6.
HCN _____________7.
CH4 _____________8.
NH3 _____________9.
CH2O _____________10. C2H2 ______________ 11. PF5
_____________ 12. SF6 |
|
Reading
Reference: Jespersen 7th Section 9.1/9.2 See Example 9.1 Page 409 See Steps page 413; See Example 9.4 Page415
Practice Exercise 9.1 page 406; Practice Exercises 9.2/9.3 Page 409; Practice
Exercises9.4-9.6 Page 415
Look
at End of Chapter Exercises #9,1-#9,7 page 458; Wrk
Review Problems #9.73-#9.82 page 460-1
Module Four II: Part O: Polarity of Molecules 1 point
Draw the dot structure for the following compounds, then state whether the molecule is polar or nonpolar:
|
_____________1. H2O _____________2. CO2 __________ _ _3.
C2H4 _______ ______4.
SO2 __________ ___5.
SO3 ________ _____6.
HCN _________ ____7.
CH4 ________ _____8.
NH3 ______________9.
CH2O ______________10. C2H2 _______________ 11. PF5
______________ 12. SF6 |
|
Reading Reference: Section 8.6; Section 9.3
See
Example 9.5 page 419
Work Practice Exercises 9.7/9.8 Page 420
Module Four II - Part P: Hybrid Orbitals of Molecules 1 points
Use the dot/stick structures
in the table to predict the hybrid orbitals that overlap to form the covalent
bond:
|
_____________1. H2O the sigma bond between either H-O _____________2. CO2 the sigma bond between either
C=O _____________2a. CO2 the pi bond between either
C=O _____________3. C2H4 the sigma bond between the
two carbons _____________3a. C2H4 the pi bond between the
two carbons _____________4. SO2 the sigma bond between S=O _____________4a. SO2 the pi bond between S=O ____________4b. SO2 the sigma bond between S-O ____________ 5. SO3
either sigma bond between S-O ____________ 5a. SO3
the sigma bond between S=O ____________ 5b. SO3 the pi bond between S=O ____________6. HCN
the single (sigma) bond between
H-C ____________6a. HCN
the sigma bond between C=N ____________6b. HCN
either pi bond between C=N ____________7. CH4
the sigma bond between any of
the H-C ____________8. NH3
the sigma bond between any of the H-N ___________ 9.
CH2O either sigma bond between
the H-C ____________9a. CH2O the sigma
bond between the C=O ____________9b. CH2O the pi
bond between the C=O ___________ __10. C2H2 the sigma bond between the C=C _________ ____10a. C2H2 either pi bond between the C=C _____________ 10b. C2H2 the sigma bond between any of the H-C |
.
|
What is difference between a Sigma (σ) and a pi (π) bond? What is a delta(Δ) Bond
Reading Reference Jespersen 7th
: Section 9.5 and 9.6
Try Practice Exercise 9.11 and 9.12 page 427; Look at Example 9.6 page 428 and
9.7 page 430-431
Try Practice Exercise 9.14 and 9.15 Page 431 Study Example
9.8Page 432 Do Practice Exercises 9.16 and 9.17 page 433, the 9.20 and 9.21
page 440