CHM 2046C Module10 Name:___________
Chapter 14 Module 10 Homework Packet
|
Module Ten: Chemical Equilibria
Chapter 14 |
Possible |
Answer |
|
|
A. Equilibrium
Constant Derivation from Reaction Rates Lecture |
1 |
|
|
|
1 |
|
||
|
C. Meaning
of the Equilibrium Constant: K; Section 14.2 & Section 14.5
p699-701 |
1 |
|
|
|
D. Meaning
of the Equilibrium Quotient: Q;
Section 14.2 p691 |
1 |
|
|
|
E. Determination of Equilibrium Constants
from Lab Data Section 14.7 p706-709 |
2 |
|
|
|
F. Determination of Equilibrium Conc from Kc Problems Section
14.8 |
2 |
|
|
|
G: La Chatelier's
Principle Problems Lecture/Section 16.7 page 788 |
2 |
|
|
|
H. Disturbing a Chemical Equilibrium Section
14.6 pages 701-706 |
1 |
|
|
|
1 |
|
||
|
K. Key Terms - Chapter 14 & More about Equations
& Eq Coefficients 14.6 |
1 |
|
|
|
Module Ten
Total: |
13 |
|
|
Part A: Equilibrium Constant Derivation 1 point
Derive the equilibrium constant expression
|
[C]c [D]d |
|
Kc
= --------------- |
|
[A]a [B]b |
from the rate expressions of the following reversible reaction:
aA
+ bB ç è cC
+ dD
Reading Reference: Lecture and Section 14.1
Part B: Equilibrium Constant Expressions 1 point
Write equilibrium constant expressions, Kc, for the following reactions they represent:
a. PCl3 (g) + Cl2 (g) ç è PCl5 (g)
Kc =
b. 2 NOCl (g) ç è 2 NO (g) + Cl2 (g)
Kc =
c. 4 HCl (g)
+ O2 (g) ç è 2 H2O (g) +
2 Cl2 (g)
Kc =
d. CS2 (g) + H2 (g) ç è CH4 (g) + H2S (g)
Kc =
e. CaCO3 (s) ç è CaO (s) + CO2 (g)
Kc =
f. NH3
(g) + H2O (l) ç è NH4
1+(aq)
+ OH1- (aq)
Kc =
Reading Reference:
Sections 14.1 and 14.2
Example 14.1 and Example 14.4
Try Practice 14.2; 14.3 p693; 14.10; 14.11 p699
Try Review Problems
14.27-14.30 page 725
Part C: The Equilibrium Constant K & Expression 1 point
Write five statements which explain what the equilibrium constant expression represents:
a)
b)
c)
d)
e)
Write five useful statements about the equilibrium constant:
a)
b)
c)
d)
e)
Reading Reference: Sections 14.2 and 14.3: Changing Dilutions P693; Add Chemical Equations p694:
Relation of Kc
to Kp page 696;
Heterogeneous Equilibrium Section 14.4
Look at Example 14.2 p695; Example 14.4 p 698;
Try Practice14.6; 14.7 page 696; Practice 14.8; 14.9 p697 Practice 14.10; 14,11 p698
Part D: Equilibrium
Constant, K & The Reaction Quotient, Q 1 point
Make
three generalizations concerning the composition of equilibrium mixtures:
a)
b)
c)
Determining a reaction quotient is useful for two reasons, state the reasons:
a)
b)
Demonstrate the Usefulness of the reaction Quotient:
The brown gas nitrogen dioxide, NO2,
will exist in equilibrium with the colorless gas, N2O4, K
= 170 at 298 K.
2 NO2 (g) ß
à N2O4 (g) K = 170
Suppose that, at a specific time, the
concentration of NO2 is 0.015M, the concentration of N2O4
is 0.025M.
a)
Is the Quotient Q larger than, or
smaller than, or equal to K?
- If
the system is not a equilibrium, in which
direction will the reaction proceed to achieve equilibrium?
Picture
Interpretation:
The Reaction A2 + B2 <====> 2 AB has an equilibrium constant Kc = 4.
The following pictures represent reaction mixtures that contain A2
molecules (red), B2 molecules (blue) and AB molecules:
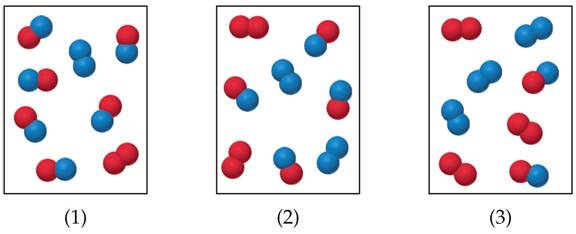
a) Which reaction mixtures is at equilibrium?
b) For the reaction mixtures that are
not at equilibrium, will the net reaction go in the forward or the reverse
direction to react equilibrium?
Reading Reference: Section
14.5
Try Practice Exercise 14.12 page 700
Try Review Problems
14.31-14.34 page 725
Part E: Experimental Determination of Equilibrium Constants 2 points
Calculate the equilibrium constant at 25oC for the reaction:
2 NOCl(g) ç è 2 NO (g) +
Cl2
when 2.00 moles of NOCl were placed in a 1.00-L flask, and the concentration of NO after equilibrium was achieved was 0.66 moles/liter.
|
|
[ ] |
[ ] |
[ ] |
|
Initial |
|
|
|
|
Change |
|
|
|
|
Equilibrium |
|
|
|
Reading Reference: Section
14.7 page 708
Look at Example 14.6 page
708
Try Practice Exercise 14.16; 14.17; 14.18 page 709
Try Review Problems
14.57-14.62 page 727
Part E: Experimental determination of Equilibrium Constants
A solution is prepared
by dissolving 0.050 mol of diiodocyclohexane, C6H10I2
in the solvent CCl4. The total solution is 1.00 Liter. When the
reaction:
C6H10I2 ß à C6H10 +
I2
has
come to equilibrium at 35oC. The concentration of the I2 is
0.035 M.
(a)What
are the concentrations of C6H10I2 and C6H10
at equilibrium?
|
|
[ ] |
[ ] |
[ ] |
|
Initial |
|
|
|
|
Change |
|
|
|
|
Equilibrium |
|
|
|
(b)
Calculate the K, the equilibrium constant.
Reading Reference: Section
14.7 page 708
Look at Example 14.6 page
708
Try Practice Exercise 14.16; 14.17; 14.18 page 709
Try Review Problems
14.57-14.62 page 727
Part F. Calculation Equilibrium Concentrations from Initial Concentrations based on Kc 2 points
At 25oC, Kc=0.090 for the reaction:
H2O (g) + Cl2O (g) ç è 2 HOCl (g)
Calculate the concentration of all species if 2.0 mole of pure H2O and 2.0 mole of pure Cl2O are placed in a 2.0 L flask and the system is allowed to come to equilibrium.
|
|
[ ] |
[ ] |
[ ] |
|
Initial |
|
|
|
|
Change |
|
|
|
|
Equilibrium |
|
|
|
Rearrange equation into the quadratic equation:
ax2
+ bx + c = 0
Then
solve by using the quadratic Formula:
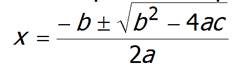
Reading Reference: Section
14.8
Look at Example 14.7 page
710
Try Practice Exercise 14.19; 14.20; page 710
Look at Example 14.8;
14.9; 14.10 Using Kc and
Initial Concentrations
page 711-718
Try Practice Exercise 14.21; 14.22;14.23 page 715-718
Try Review Problems
14.63-14.82 page 727-728
Part F. Calculations based on Kc continued
The reaction
. N2 (g) +
O2 (g) ç è 2 NO
(g)
contributes to air pollution whenever fuel is burned at high temperatures and high pressure in an automobile gasoline engine. At 1500 K, K= 1.0 x 10 -5. Suppose a sample of air has
[N2] = 0.80 M and [O2] = 0.20 M before any reaction occurs. Calculate the equilibrium concentrations of the reactants and the products after the mixtures reaches 1500 K?
|
|
[ ] |
[ ] |
[ ] |
|
Initial |
|
|
|
|
Change |
|
|
|
|
Equilibrium |
|
|
|
You may use assumptions I this calculation, show and prove assumption
Reading Reference: Section
14.8
Look at Example 14.10
Using Kc and Initial Concentrations page
716-718
Try Practice Exercise 14.23; 14.24 page 718
Try Review Problems
14.63-14.82 page 727-728
Part G: LaChatelier’s Principle 2 points
Consider the isomerism of butane with an equilibrium constant of Kc = 2.5.
CH3
|
CH3-CH2-CH2-CH3 (n-butane) ç è CH3-CH-CH3 (isobutane)
The system is originally at equilibrium with [n-butane]=1.0 M and [isobutane]=2.5M.
(a) If 0.50 moles/liter of isobutane is suddenly added and the system shifts to a new equilibrium position, what is the equilibrium concentration of each gas?
|
|
[ ] |
[ ] |
|
Initial |
|
|
|
Change |
|
|
|
Equilibrium |
|
|
Reading Reference:
None--Lecture
No Practice Problems
Part G: LaChatelier’s Principle continued
Carbonyl bromide decomposes to carbon monoxide and bromine gas.
COBr2 (g) ß à CO (g)
+ Br2 (g)
K is 0.190 at 73oC. Suppose you placed 0.500 moles of COBr2 in a 1.00-L flask and heated it to 73oC. After equilibrium had been achieved, you added an additional 3.00 mol of CO.
Calculate the equilibrium concentration of each component before the CO is added:
|
|
[ ] |
[ ] |
[ ] |
|
Initial |
|
|
|
|
Change |
|
|
|
|
Equilibrium |
|
|
|
How is the equilibrium mixture affected by adding more CO?
When equilibrium is reestablished, what are the new equilibrium concentrations of each component?
How has the addition of CO affected the concentration of COBr2 that decomposed?
Part H: LaChatelier’s
Principle Questions 1 point
Based on the following Equations, use the choices to describe the equilibrium shifts, if any, when the following conditions are changed:
a. shifts left b. shifts right c.
No effect
4 HCL (g) +
O2 (g) ç è 2 H2O (g) + 2
CL2 (g)
_____1. 1 mole of Chlorine gas is injected after
equilibrium is reached
_____2. Water is removed after equilibrium is
reached
_____3.
Additional Heat is applied to system after equilibrium is reached,
(the reaction is exothermic)
_____4. Nitrogen gas is injected into the vessel
after equilibrium is reached
_____5. Additional Oxygen gas is injected after
equilibrium is reached
_____6. Increasing the
size of the vessel from 1 to 3 liters after eq is
reached
PCL5
çè PCl3 + CL2
_____7. Chlorine gas is injected into the vessel
_____8. Additional Heat is added to the system after
equilibrium is achieved
(the
reaction is endothermic)
_____9. Nitrogen gas is injected into the vessle
_____10. The gases are compressed into a vessel ½
half the original size
after equilibrium is achieved.
Reading Reference: Section
14.6
Look at Example14.5 page 705
Try Practice: 14.14;14.15 page 706
Review Problems: 14.53-14.56 pages
726-727
Part
I: Discussion Questions 1 point
1. What
is dynamic equilibrium?
2. What
is true about the initial rate of
the forward and reverse reactions in a system where only reactants are present?
- a. What is true about the rates of the
forward and reverse reactions at
equilibrium?
b. What happens to the value of K when the reaction is
reversed?
c. What happens to the value of K when
the temperature is changed for the reaction?
4. Your
textbook introduces the concept of equilibrium by noting that no reaction goes fully to
completion. What does this imply about
the reverse reaction?
5.
Discuss the relationship between the magnitude of the equilibrium constants as to whether the forward or the reverse reaction is favored.
6. What
effect does adding a catalyst on the
concentrations of the reactants and products after equilibrium is established.
7. What disturbances are possible
for a system in equilibrium?
8. When is it reasonable to
neglect the change in the concentration in the denominator of an equilibrium
constant expression when solving for the equilibrium concentrations from the
initial concentrations.. (When does 1.0 M – x ~ 1.0?)
Also see review questions:
#14.1-#14.26 pages 724-725
Part K: Vocabulary / More Equilibrium Shifts Chapter 14 (1 point)
__________________ 1. If all the reactants and the products are in the same phase (such as
gases), then the we say the system is in _______________equilibrium.
__________________ 2. If at least one of the reactants or one of the products is not in the same
phase, then we say the system is in __________________ equilibrium.
__________________ 3. The state where the concentrations of the reactants and the products
remain constant with time , it is said that the system has reached a state
of ___________equilibrium.
__________________ 4. The _______ _________ Q is obtained by by applying the law of mass
action using the initial concentrations of both the reactants and the
products instead of the equilibrium concentration so as to determine the
direction of the equilibrium shift.
___________________5. ____________ ________________ states that if a change is imposed
on a system in
equilibrium , the position of the equilibrium will shift in
a direction that tends
to reduce that change.
Predict the effect on each of the following changes to the system on the direction of equilibrium:
a. Moves to the Right
b. Moves
to the Left
c. Has no effect/does not move left or right
The combination of hydrogen gas and oxygen gas to give water
vapor can be expressed by
2 H2 (g) + O2 (g) çè 2 H2O (g)
_________6. Water is removed as it is being generated
_________ 7. Additional H2 is added
_________ 8. The system is cooled (assuming the reaction is exothermic)
_________ 9. The container is made larger
_________10. Nitrogen gas (which does not react with neither reactants nor products) is injected
into the cylinder causing a change in the pressure, Volume of vessel remains the
same.