Copyright © 2008 Pearson
Prentice Hall, Inc.
Chapter 4/36

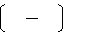
Oxidation-Reduction (Redox) Reactions
b)Oxygen
usually has an oxidation number of -2.
H
O
1-
-2
+1
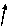
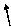
H
H
Ca
-1
-1
+2

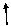

3.An atom in a
polyatomic ion or in a molecular compound usually has the same oxidation number it would have if it were a monatomic ion.
a)Hydrogen
can be either +1 or -1.
O
H
O

-1
+1
-1


H
H
O
+1
+1
-2


H

+1
