Copyright © 2008 Pearson
Prentice Hall, Inc.
Chapter 7/18

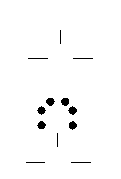
Electron-Dot Structures of Polyatomic
Molecules
Draw an
electron-dot structure for CH2O.
4 +
2(1) + 6 = 12 valence electrons
Step
3:
Step
1:
Step
2:
H
C
O
H
H
C
O
H
Step
5:

H
C
O
H
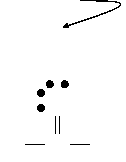
H
C
O
H