Copyright © 2008 Pearson
Prentice Hall, Inc.
Chapter 7/23

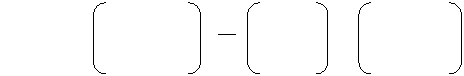
Formal Charges
Formal
Charge
# of
valence e-
in free atom
-
2
1
-
# of
nonbonding
e-
# of
bonding
e-
=
Calculate
the formal charge on each atom in O3.
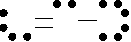
O
O
O
6
- (2) - 6 = -1

1
2
6 - (4) - 4 = 0
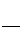
1
2
6
- (6) - 2 = +1

1
2
