Copyright © 2008 Pearson
Prentice Hall, Inc.
Chapter 7/53

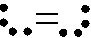
Molecular Orbital Theory: Other
Diatomic Molecules
Oxygen, O2, is predicted to be diamagnetic by electron-dot structures and valence bond
theory.
However, it is known to be paramagnetic.
O2
O
O
Diamagnetic: All
electrons are spin-paired. It is weekly repelled by magnetic fields.
Paramagnetic: There is at least one unpaired electron. It is weakly attracted by magnetic fields.